1. Introduction
The global population is estimated to increase steadily, reaching approximately 2.4 billion more people by 2050, necessitating a 70% increase in food resources (Aiking, 2011). As the growing global population, the global demand protein sources is estimated to increase correspondingly (Mintah et al., 2020). The conventional sources of protein are primarily derived from animals, such as cows, pigs, and chickens; however, growing concerns about economic and environmental issues are driving demand for alternative protein sources (Gravel and Doyen, 2020). Edible insect proteins are recognized to resolve these issues, providing nutritional benefits, emitting fewer greenhouse gases, and being inexpensive to farm (Cho et al., 2025a; Gravel and Doyen, 2020; Oonincx et al., 2010; Rumpold and Schlüter, 2013).
Among edible insects, Zophobas morio (ZM), known as the super mealworm, a member of the darkling beetle family, has gained a lot of attention for its rich nutrients (Rumbos and Athanassiou, 2021). This species has a similar shape to Tenebrio molitor and originated in South and Central America (Cho et al., 2025b; Park et al., 2013). In addition, numerous studies have revealed that ZM protein exhibits outstanding functional properties, including protein digestibility, emulsification, and foaming capacities (Kim et al., 2015; Kulma et al., 2020; Zielińska, 2022). Moreover, recent studies have demonstrated that its protein isolates or hydrolysates have considerable biological activity, such as antioxidants and anti-inflammatory activities, thereby increasing its applicability in not only the food but also the pharmaceutical industries.
Macrophages, which are the most representative immune cells, prevent the invasion of foreign antigens and regulate the immune system (Martinez et al., 2008). They are activated by external stimuli and have enhanced phagocytosis activity that nonspecifically recognizes and removes invading antigens, and function to regulate the immune system by expressing immune modulators (Mosser and Edwards, 2008). If macrophages are not properly activated, immune deficiencies may occur, impairing the ability to fend off invading pathogens and thereby disrupting homeostasis (Mosser et al., 2021; Wynn et al., 2013). Therefore, substances capable of macrophage activation and thereby enhancing immunological responses are referred to as immune-enhancing agents (Bi et al., 2018; Kim et al., 2017; Li et al., 2017; Park et al., 2024a).
The conventional water extraction method is simple, easily applicable, low-cost and cost-effective process (Pan et al., 2022). Thus, recent studies have extensively investigated the physiological activities of edible insect water extracts, such as the antioxidant and immune-enhancing capacities of Tenebrio molitor, Protaetia breviarsis, and Allomyrina dichotoma larvae (Kim et al., 2023; Park et al., 2023; Park et al., 2024a; Shin et al., 2023). Despite the primary focus of research on the biological activities of ZM protein, this study sought to investigate the functional profile of the Zophobas morio water extract (ZMWE) by characterizing its chemical compositions, structural, antioxidant, and immune-enhancing properties.
2. Materials and methods
ZM was purchased from a cricket farm (Suwon, Korea). The ZM was properly cleaned with tap water and dehydrated in a dry oven (JSOF-100, Gongju, Korea) at 60°C (2 days). Following drying, the sample was ground up using a grinder (RT-04, Mill powder, Tainan, Taiwan) for 30 s and kept at −18°C until further experiments.
Water extraction was accomplished using the prior approach with modest adjustments (Park et al., 2024b). The ground ZM was defatted two times following our previous study (Cho et al., 2025b). Briefly, the defatted sample was mixed with distilled water to a final concentration of 10 mg/mL and heated at 60°C for 6 h in a water bath (GO-90W, Jeio Tech). After cooling, the extract was centrifuged at 6,000 ×g for 10 min at 4°C (Laborgene 1248R, Labogene Co., Ltd., Daejeon, Korea) to collect the supernatant. Subsequently, the collected supernatant was filtered using an 8 μm filter (Whatman No. 2, Whatman International Ltd., Maidstone, UK) under a vacuum condition. The whole extraction process was conducted once. Then, the final product was freeze-dried (FDS8518, Ilsin 115 BioBase Co. Ltd., Dongducheon, Korea). The dried ZMWE was located in a zip package at −18°C. The extraction yield of ZMWE was determined as 9.98±0.02% based on the defatted powder weight.
The protein content of ZMWE was calculated using the Kjeldahl method according to the Association of Official Analytical Chemists (AOAC, 1990). The total carbohydrate content was analyzed as described by Dubois et al. (1956). The calibration curve of the standard glucose (12.5-100 μg/mL) was used. The moisture and ash contents were recorded according to the standard methods of the AACC (AACC, 2000). The total phenol content (TPC) in ZMWE was determined as described by Olawuyi et al. (2020b), with results expressed as mg Gallic Acid Equivalent (GAE)/g dry weight (DW).
FT-IR analysis of ZMWE was investigated using an FT-IR spectrophotometer (Frontier, PerkinElmer, MA, USA) by Cho et al. (2025a). FT-IR spectra were recorded from 400 to 4,000 cm−1 with a resolution of 4 cm−1.
The sample solution (1.25-10 mg/mL) was prepared by mixing ZMWE with distilled water. Then, the mixture was centrifuged at 4,000 ×g for 10 min at 4°C, and the collected supernatant was used for antioxidant analyses.
The ABTS radical scavenging assay was evaluated as described by Baek et al. (2024). Briefly, 7 mM ABTS and 2.45 mM potassium persulfate in distilled water were transferred to the flask and covered with foil. The solution was stirred continuously using a magnetic bar for 16 h to generate the ABTS reagent. After that, 50 μL of ZMWE solutions were mixed with 950 μL of ABTS reagent and incubated at 25°C for 30 min. The absorbance of the mixture was recorded at 734 nm using an ultraviolet (UV) spectrophotometer (UV-2550, Shimadzu Corp., Kyoto, Japan). The ABTS radical scavenging capacity (RSC, %) was calculated using the following equation (1).
Where A0 and A1 denoted the absorbance numbers for the control and ZMWE, respectively.
The DPPH radical scavenging assay was evaluated as described by Aliya et al. (2025). In brief, 3.9 mg of DPPH in 95% ethanol (100 mL) was stirred with continuous magnetic stirring for 2 h at 25°C to produce the DPPH reagent. For the analysis, 100 μL of ZMWE solutions were combined with 900 μL of DPPH reagent and then reacted at 25°C for 30 min. The DPPH radical scavenging activity (%) was measured by comparing the sample absorbance to that of a control using an UV spectrophotometer at 517 nm. The DPPH radical scavenging capacity (RSC, %) was determined as follows:
Where A0 and A1 denoted the absorbance numbers for the control and ZMWE, respectively.
The FRAP of ZMWE was determined as described in a previous study (Park and Lee, 2021). 100 μL of ZMWE solutions were incubated with 900 μL of FRAP reagent for 30 min at 25°C. Thereafter, the absorbance of the mixture was analyzed by a UV spectrophotometer at 593 nm. Ascorbic acid, used as a standard, was within the range of 5 to 50 μg/mL, and the results were expressed as mg ascorbic acid equivalent (AAE)/g dry weight (DW).
RAW264.7 mouse macrophage cell line was acquired from Korea Cell Line Bank (Seoul, Korea), and maintained in Dulbecco’s Modified Eagle Medium (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin under humidified atmosphere at 37°C with 5% CO2. In this study, a lipopolysaccharide (LPS) treatment group was added in addition to the experimental groups as a positive control to confirm the normal experimental conditions.
Cell counting kit-8 (CCK-8) assay was utilized to analyze the effect of ZMWE on the RAW264.7 cell viability. RAW264.7 cells (5×103 cells/well) were seeded and exposed to ZMWE for 24 h. The CCK-8 solution was then added in 10 μL for 1 h, and the absorbance was analyzed at an optical density of 450 nm using an Infinite f50 nanoplate reader (TECAN, Männedorf, Switzerland). The cell viability was calculated as follows:
Nitric oxide production was analyzed from the cell supernatants using the Griess reagent system (Promega, Madison, WI, USA). RAW264.7 cells (1×105 cells/well) were seeded and exposed to ZMWE or LPS (1 μg/mL) for 24 h. Subsequently, 50 μl of cell supernatants from each experimental group were sequentially added with same volume of sulfanilamide solution and N-(1-napthyl) ethylenediamine solution at 10 min intervals. The absorbance was analyzed at an optical density of 570 nm using an Infinite f50 nanoplate reader (TECAN).
The samples were prepared with RIPA buffer (Biosesang, Korea) containing protease/phosphatase inhibitors. The proteins were separated utilizing sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), followed by membrane transfer procedure. Subsequently, the membrane was blocked using 5% skim milk in Tris-buffered saline containing 0.1% Tween 20 (TBST). After reacting overnight with the primary antibodies (Table S1) and rinsing with TBST, the membrane reacted with horseradish peroxidase-conjugated secondary antibodies. The membranes were rinsed with TBST before detecting, and the detection was performed utilizing the enhanced chemiluminescence detection kit (Cytiva, Marlborough, MA, USA). The density of the western blot bands was calculated using Image J and normalized relative to the LPS-treated group.
The secretion of prostaglandin E2 (PGE2), tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) from cell supernatants were quantified using ELISA kits (R&D Systems, Minneapolis, MN, USA).
Attune Acoustic Focusing Cytometer (Thermofisher Scientific, Waltham, MA, USA) was utilized for flow cytometry analysis.
All experimental groups were allowed to react with 20 μM of DCF-DA and incubated for 30 min. The cells were rinsed, and the intracellular fluorescence was analyzed by flow cytometry with excitation/emission (Ex/Em) wavelength of 488/530±15 nm.
Before staining, the nonspecific binding was prevented by exposing to anti-cluster of differentiation (CD) 16/32 (eBioscience) for 20 min at 4°C. Subsequently, the cells were stained with CD80 Monoclonal Antibody-PE (eBioscience) or major histocompatibility complex (MHC) class II Monoclonal Antibody-PE (eBioscience) for 30 min at 4°C. The fluorescence was analyzed by flow cytometry with Ex/Em wavelength of 488/574±13 nm.
Vybrant™ Phagocytosis Assay Kit (Thermofisher Scientific) was used to perform phagocytosis activity in compliance with our previous study (Park et al., 2024a). Cells were exposed to Fluorescein-labeled Escherichia coli (E. coli) K-12 bioparticles for 120 min, following the removal of the bioparticles, and trypan blue was added for 1 min. The cells were recovered, and the fluorescence was analyzed by flow cytometry with Ex/Em wavelength of 488/530±15 nm. The phagocytosis effect was calculated using the following equation (4):
The data were presented as mean±standard deviation (SD) analyzed using a student t-test or one-way ANOVA followed by Duncan’s Multiple Range Test for statistical significance (p<0.05). Statistical analysis was performed using IBM SPSS Statistics (version 22.0, IBM Corp., USA). All experiments were repeated in triplicate.
3. Result and discussion
The chemical compositions of ZMWE are presented in Table 1. The protein, carbohydrate, moisture, ash, and polyphenol were 58.24%, 20.80%, 7.94%, 15.31%, and 0.91 mg GAE/g DW, respectively. These results confirm that the major compounds of ZMWE are water-soluble proteins and carbohydrates, with relatively lower total phenol content. Similarly, Jeong et al. (2021) reported that the water extract obtained from Tenebrio molitor had a protein, carbohydrate, and ash content of 56.53%, 20.93%, and 12.88%, respectively. In addition, Zhang et al. (2023) also observed that a superior protein content of 53.81% in the water extract from Protaetia brevitarsis larvae. Consequently, the chemical composition of ZMWE is comparable to that of previous studies. In addition, given these higher nutritional values, it can be an appropriate material for functional foods.
| Moisture (%) | Protein (%) | Ash (%) | Carbohydrates (%) | Total phenol content (mg GAE/g DW)1) | |
|---|---|---|---|---|---|
| ZMWE | 7.94±1.142) | 58.24±0.34 | 15.31±5.85 | 20.80±0.45 | 0.91±0.02 |
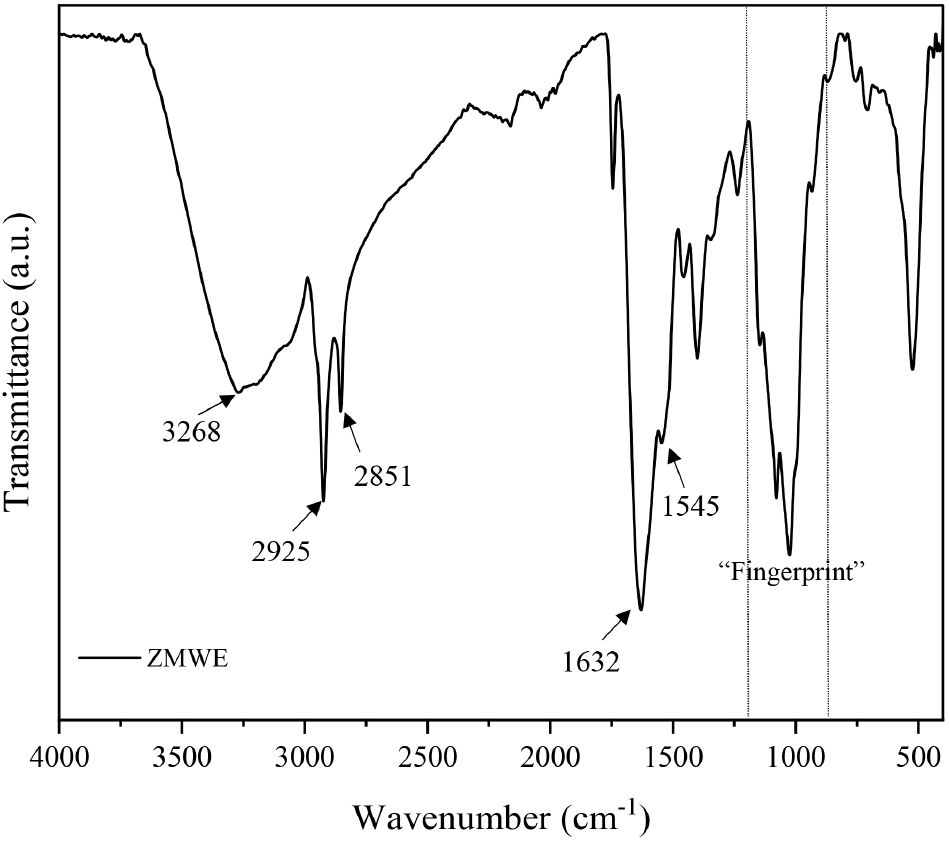
FT-IR analysis was conducted to confirm the structural properties of ZMWE. The spectra of ZMWE from 400 to 4,000 cm−1 are presented on Fig. 1. FT-IR spectra exhibited several characteristic peaks. The wide and sharp peaks at 3,000-3,600 cm−1 and 2,851-2,925 cm−1 are associated with the O-H bond as well as CH2 and CH3 stretching vibration, respectively, indicating the typical absorption peaks of edible insect protein or polysaccharide (Cho et al., 2025c; Park et al., 2024b). The prominent peak at 1,631 cm−1 and small peak at 1,545 cm−1 can be assigned to the stretching vibration of C=O and N-H of amide I and II (protein) (Glassford et al., 2013). Meanwhile, the “fingerprint” band areas are observed at 800-1,200 cm−1, indicating the presence of carbohydrate in the ZMWE (Olawuyi et al., 2020a). Thus, the result of the FT-IR band confirms the coexistence of protein and polysaccharide in the ZMWE.
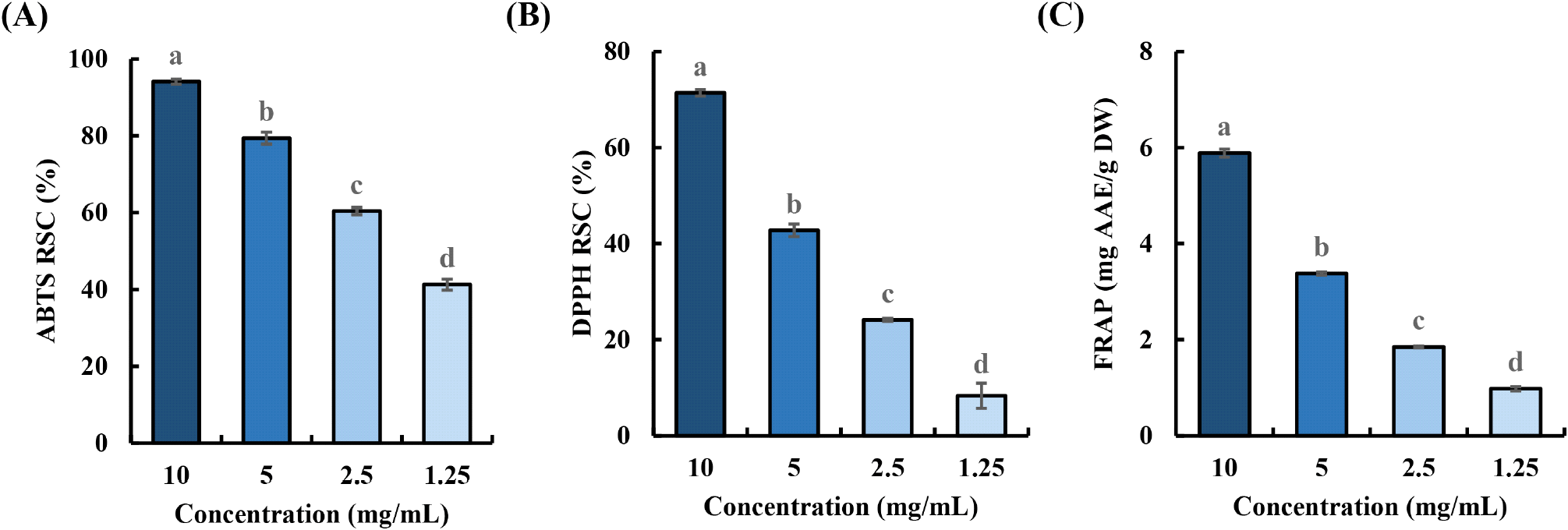
The antioxidant properties of ZMWE were analyzed using the ABTS and DPPH radical scavenging capacity, as well as the FRAP assay, at concentrations ranging from 1.25 to 10 mg/mL. As shown in Fig. 2, ZMWE exhibits dose-dependent response, and their calculated ABTS IC50 and DPPH IC50 values are 1.39 and 6.61 mg/mL, respectively. Similarly, in the FRAP assay, ZMWE showed the highest ferric reducing power of 5.89 mg AAE/g. The antioxidant properties of ZMWE can be attributed to antioxidant amino acids or polysaccharides, which have the ability to neutralize free radicals (Lai et al., 2010; Samaranayaka and Li-Chan, 2011). For example, amino acids, including phenylalanine, methionine, and histidine, are known for exhibiting antioxidant activities due to their aromatic rings or sulfur groups. Meanwhile, compositional monosaccharides such as uronic acid, which can supply hydrogen ions, are reported to be related to the display of antioxidant properties (Cho et al., 2025a; Olawuyi et al., 2020a). In addition, these findings are consistent with a previous study of antioxidant capacities of water extract from Tenebrio molitor (Kim et al., 2024). Therefore, these findings demonstrate that ZMWE possesses antioxidant properties.
Activated macrophages express key immune modulators, such as inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), and pro-inflammatory cytokines. iNOS produces nitric oxide during the metabolism of L-arginine to L-citrulline, while COX-2 biosynthesizes PGE2 from arachidonic acid (Billack, 2006; Hinz et al., 2000). Both nitric oxide and PGE2 play pivotal roles in immune response. Nitric oxide regulates blood pressure, contributes to wound healing, eliminates foreign microbes, and has antiviral capacity. Meanwhile, PGE2 facilitates macrophage migration and enhances the chemotactic responses of various chemokines (Billack, 2006; Nathan and Hibbs, 1991; Tajima et al., 2008). Also, macrophages expressed pro-inflammatory cytokines represented by TNF-α and IL-6 play crucial roles in regulating immune responses, activating other immune cells, and eliminating pathogens (Murray, 2017; Parameswaran and Patial, 2010; Stevens et al., 1992; Van Snick, 1990). These immune modulators expression in macrophages is used to verify the immune-enhancing properties of bioactive substances (Wu et al., 2018).
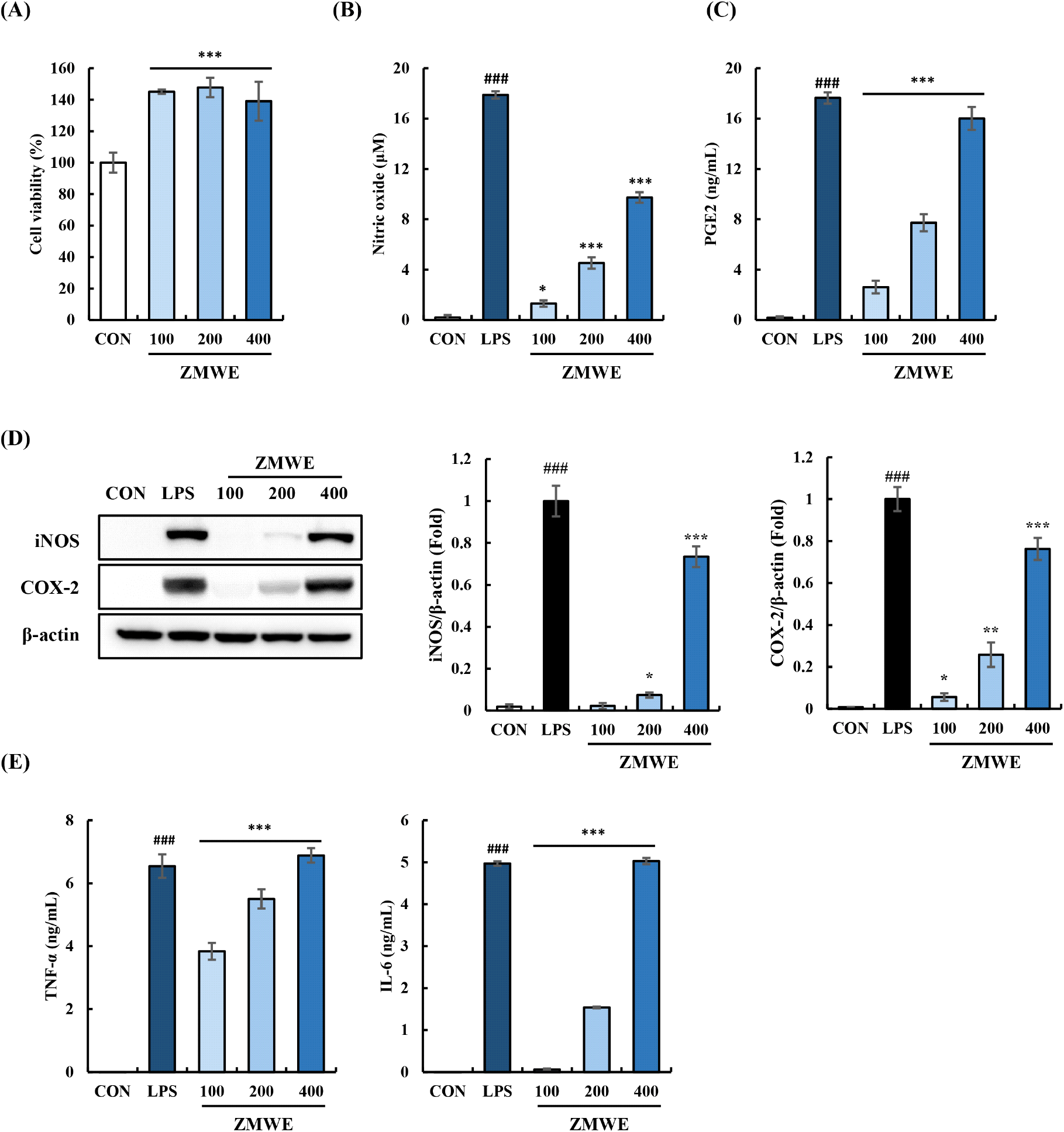
First, the cell viability experiment was performed at various concentrations (100-400 μg/mL) to verify the cytotoxicity of ZMWE in RAW264.7 cells. (Fig. 3A). Under the tested doses (100-400 μg/mL), ZMWE significantly increased the cell viability (139.08-145.12%) compared to the non-treatment group (100±6.36%), suggesting that ZMWE induced macrophage proliferation. Increased proliferation of macrophages can contribute to the immune-enhancing activities by effectively improving non-specific immunity, boosting the effectiveness of pathogen elimination (Fang et al., 2017). Then, the effect of ZMWE on the immune modulators’ expression is shown in Fig. 3B-3D. ZMWE treatment significantly increased the secretion of nitric oxide and PGE2, following the iNOS and COX-2 expression (Fig. 3B and 3C). Notably, ZMWE treatment induced higher secretion of nitric oxide (1.30-9.73 μM) and PGE2 (2.62-16.02 ng/mL) in line with the iNOS and COX-2 expression (0.02-0.73 folds and 0.06-0.76 folds). Accordingly, as shown in Fig. 3D, TNF-α and IL-6 secretion (3.84-6.88 ng/mL and 0.06-5.03 ng/mL) was observed in the ZMWE treatment groups. To summarize, these results highlighted the role of ZMWE on the immune modulators’ expression in macrophages, suggesting the immune-enhancing property of ZMWE.
ROS are synthesized upon macrophage activation, which contributes not only to eliminating the invading pathogens but also to regulating the homeostasis function of macrophages (Tan et al., 2016). Previous studies have highlighted the significance of ROS, with increased ROS production in macrophages being recognized as an immune-enhancing property (Canton et al., 2021). DCF-DA assay was performed to analyze the effect of ZMWE fractions on ROS production, and the result is shown in Fig. 4 and Fig. S1A. DCF-DA+ area significantly increased in ZMWE in a dose-dependent manner (47.27-51.96%), suggesting that ZMWE upregulated intracellular ROS production in RAW264.7 cells compared to the non-treatment group (10.01±0.28%). Kim et al. (2025) have also demonstrated the immune-enhancing activity of Nelumbo nucifera root extracts, observing the increased ROS production in macrophages. Likewise, our data suggests the increase in ROS production by ZMWE treatment in macrophages, supporting the immune-enhancing activity of ZMWE.
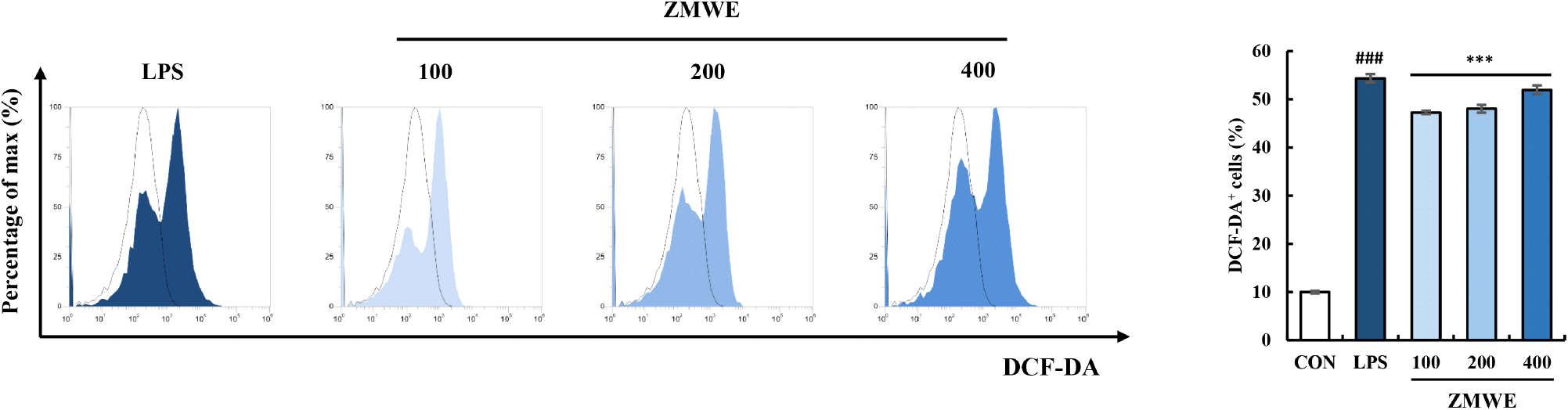
The results of the antioxidant activity assays (ABTS, DPPH and FRAP) and intracellular ROS production assay (DCF-DA) appeared to present conflicting patterns. The antioxidant activity assay aimed to determine the potential to provide electrons or stabilize free radicals, which demonstrates the potential to decrease ROS in the extracellular environment. Meanwhile, the increased ROS production in macrophages should be discussed not as oxidative stress, but as a key step in physiological immune responses. When the macrophage detects antigens, it triggers a temporary respiratory burst that generates ROS, which serves as an essential defense mechanism to kill ingested pathogens but also upregulates the macrophage activation signaling pathway (Canton et al., 2021; Geum et al., 2020). Therefore, the result of intracellular ROS production in ZMWE-treated RAW264.7 cells demonstrates the role of ROS as an immune-modulator that promotes immune responses of macrophages. For further study, it is necessary to confirm whether the ROS production was transient and occurred at controllable levels.
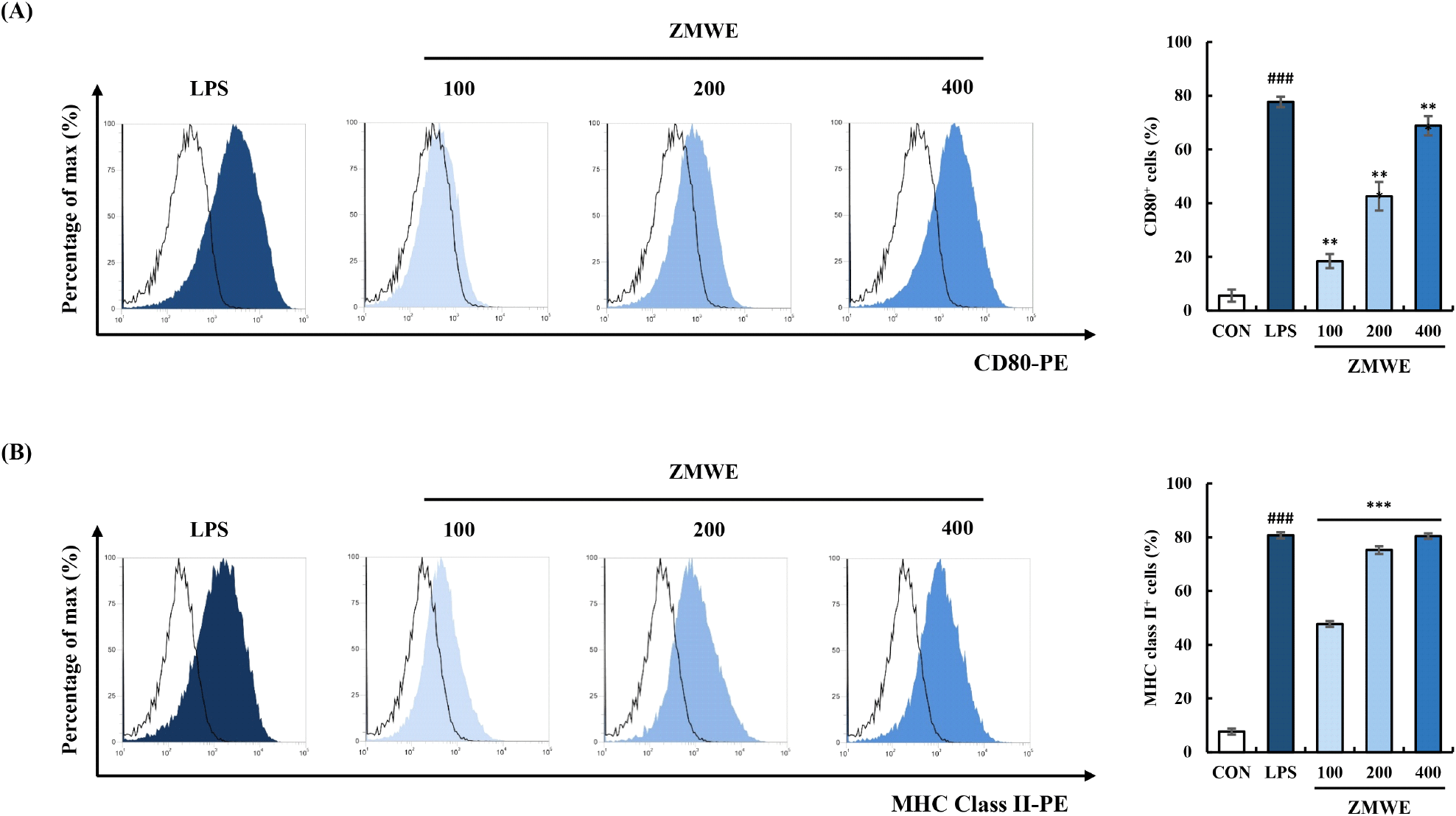
CD80 and MHC class II, expressed on macrophages, interact with T cells to modulate their activation (Togno-Peirce et al., 2013). Their increased expression in macrophages not only indicates an activation phenotype of macrophages but also enhances T cell-mediated immune responses (Park et al., 2024b; Xiong et al., 2024; Xu et al., 2021). Fig. 5 and Fig. S1B-S1C present the effect of ZMWE fractions on the expression of macrophage surface activation biomarkers. A significant increase in CD80 and MHC class II expressions on the surface of RAW264.7 cells was observed by ZMWE (18.41-68.85% and 47.78-80.43%) treatment compared to the control group (5.64±2.27% and 7.75±1.13%). The results indicated that the ZMWE enhanced the M1 macrophage polarization phenotypes of RAW264.7 cells.
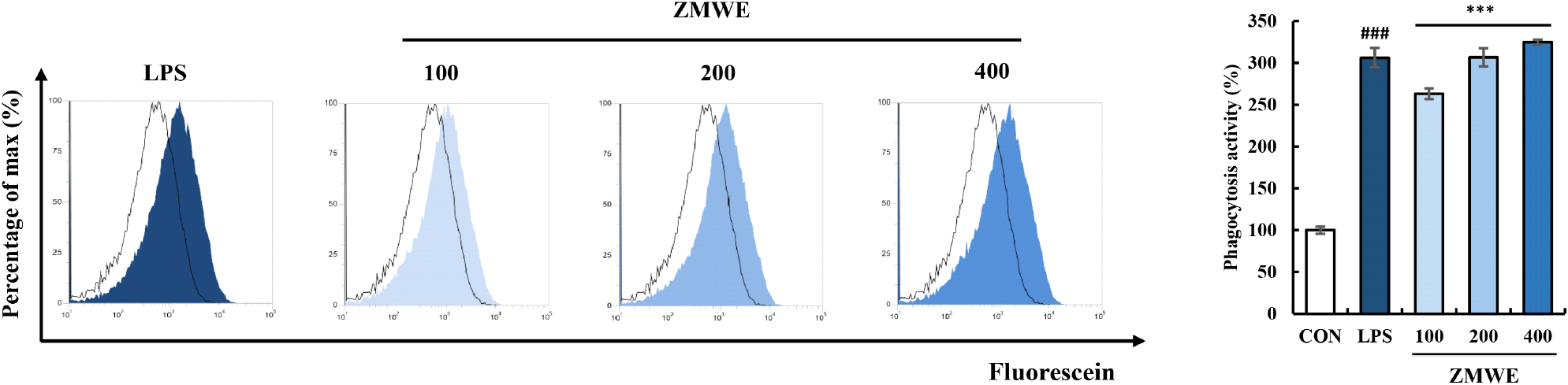
Phagocytosis activity is one of the well-known properties of macrophages in innate immunity, which nonspecifically recognizes and engulfs invading antigens (Aderem and Underhill, 1999). Increased phagocytosis activity has been suggested as an immune-enhancing property of macrophages in previous studies (Park et al., 2015; Wang et al., 2022). The effect of ZMWE on the phagocytosis activity was confirmed by measuring the contents of Fluorescein-labeled E. coli bioparticles engulfed inside the cells, which is shown in Fig. 6 and Fig. S1D. Compared to control groups (16.58±0.71%), increased engulfed bioparticles were observed in ZMWE treatment groups (43.64-53.87%), and when normalized, phagocytosis activity was increased to 263.22-324.91% for ZMWE treatment compared to the non-treatment group (100±2.94%). This result suggests that ZMWE fractions improve phagocytosis activity in macrophages.
The macrophage activation capacity of bioactive compounds derived from insects has been reported in numerous studies. Lee et al. (2023) and Park et al. (2024b) have suggested that the polysaccharide and protein derived from Protaetia brevitarsis larvae are the active ingredients for macrophage activation. Also, Ali et al. (2019) have reported the macrophage-activation properties of the polysaccharide derived from Hermetia illucens, known as dipterose. In addition, Guo et al. (2024) isolated peptides from silkworm pupa protein hydrolysates and reported their macrophage-activating properties. These studies highlight the potential of insects as sources of immune-enhancing compounds that induce activation of macrophages and suggest that the active components are carbohydrates or proteins.
4. Conclusions
In the present study, a water extract was prepared from Zophobas morio, a recognized new eco-friendly material, and its biological activities were investigated to increase industrial applicability. The major components of ZMWE are protein and carbohydrate, as determined by chemical composition and FT-IR analyses. The antioxidant properties of ZMWE were revealed by reducing ABTS and DPPH free radicals and the FRAP assay. In addition, the Griess reagent assay and ELISA confirmed the effects of ZMWE on the secretion of nitric oxide, PGE2, and pro-inflammatory cytokines (TNF-α and IL-6) in macrophages. Moreover, flow cytometry confirmed the effects of ZMWE on ROS production, phagocytosis activity, and expression of m1 macrophage surface markers (CD80 and MHC class II). These results suggest the capacity of ZMWE to activate macrophages. These findings suggest its potential as an antioxidant agent or functional ingredient. However, further investigation is needed to unveil which component of ZMWE is the key activator that contributes to its antioxidant and immune-enhancing properties, to elucidate the underlying mechanism.