1. Introduction
Groundnuts, or peanuts (Arachis hypogaea L.), are a significant upland crop worldwide, particularly in tropical and subtropical areas (Krishna et al., 2015). Peanuts are an important source of raw materials for the manufacturing of vegetable oil because of their high oil content (40-60% of seed weight) (Akgül and Tozluoğlu, 2008). Because it contains unsaturated fatty acids, vitamin E, and antioxidant components, peanut oil has gained popularity as a culinary ingredient and is valued for its nutritional content (Toomer, 2018).
A. hypogaea varieties differ in their economic uses due to variations in their genetic and biochemical characteristics. Nowadays, a wide range of peanut types are cultivated in Vietnam for a number of uses, including food, oil production, and animal feed (Toomer, 2020).
Understanding the physical properties of pressed oils derived from different peanut cultivars is essential, particularly in regions with unique agro-ecological conditions. Despite this importance, limited research has been devoted to characterizing pressed peanut oils from varieties grown in Northwest Vietnam—a region distinguished by specific soil types and climatic conditions. A comprehensive investigation into these oils will help clarify inter-cultivar differences in composition and quality, contribute to scientific knowledge, and build a valuable database to support local farmers and the peanut oil processing industry.
Therefore, this study aimed to evaluate and compare the physical attributes, chemical profiles, oxidative stability, and antibacterial properties of pressed oils extracted from three peanut varieties cultivated in Northwest Vietnam. The findings are expected to shed light on the functional potential of these oils and inform future industrial applications.
2. Materials and methods
This study utilized three different peanut oil varieties (A. hypogaea L.), including “Lac Van” (SPO) and “Lac Den” (BPO) from Lao Cai, Vietnam, and “Lac Do” (RPO) from Dien Bien, Vietnam. The oil extraction process was meticulously conducted at 160°C using a 430 W oil press (Hakawa, model ED43, Guangdong, China). The outlet oil temperature during pressing ranged from approximately 50 to 60°C. After extraction, the crude oil was centrifuged (NEUATION, model iFuge D06, Ahmedabad, India) at 1,400 ×g for 10 min to remove residue, then sealed in bottles and stored at room temperature, ready for further chemical composition analysis.
This study utilized two standard Gram-negative bacterial strains, Escherichia coli (ATCC 25922) and Salmonella Enteritidis (ATCC 13076), provided by the Institute of Biotechnology and Food Technology at the Industrial University of Ho Chi Minh City.
The chemicals used in the study included 2,2-diphenyl-1-picrylhydrazyl (DPPH, Sigma, St. Louis, MO, USA) and dimethyl sulfoxide (DMSO, Nanjing, China). Additionally, the culture and antibacterial testing media, such as Mueller-Hinton agar (HiMedia, Thane, India) and nutrient broth (HiMedia, Thane, India), as well as other chemicals, were of analytical grade.
The colorimeter (Konica Minolta, model CR-400, Osaka, Japan) was used to measure the oil color. The following is an interpretation of the significant parameters in the CIE color space model: L* represents lightness, a* represents green or red, and b* represents blue or yellow.
Using an S63 spindle spinning at 100 rpm (unit of measure in centipoise, cP), the viscosity was measured with a viscometer (Brookfield Ametek, model LVDV-E, Middleborough, MA, USA).
The percentage of oil weight in relation to seed weight (%, v/w) was used to calculate the recovery yield. The physical properties of the oils were determined according to the International Organization for Standardization (ISO), including density (ISO 6883, 2017), acid value (ISO 660, 2020), saponification value (ISO 3657, 2023), and peroxide value (ISO 3960, 2017); while ester value was calculated based on the study of Jain (2023).
Fatty acid composition was analyzed by gas chromatography-mass spectrometry (GC-MS) using a Shimadzu GC-2030 gas chromatograph coupled to a QP2020 mass spectrometer (Shimadzu Corporation, Kyoto, Japan) equipped with an Rxi-5MS capillary column (30 m×0.25 mm i.d.×0.25 μm film thickness). Column temperature: 50°C for 4 min, then increased to 80°C at a rate of 2°C/min, 150°C at a rate of 5°C/min, 200°C at a rate of 10°C/min, and 300°C at a rate of 20°C/min, and held for 3 min. The ion chamber’s temperature was 230°C. Helium (He) was used as the carrier gas at a flow rate of 1.69 mL/min. Split the stream at a 1:10 ratio (split) with a head pressure of 100 kPa. The fatty acids were identified by comparing the retention times (RT) of a standard mixture to those of the fatty acids’ spectra with those in the NIST spectral library.
Antioxidant capacity (AC) was assessed using free radical scavenging activity (RSA) using the DPPH assay, as described by Quyen and Quoc (2024), with minor changes. To achieve various concentrations, the oil was dissolved in 96% ethanol. Then, 0.3 mL of solution was combined with 2.7 mL of 0.1 mM DPPH solution and incubated at room temperature in the dark for 30 minutes. A spectrophotometer (Thermo Fisher Scientific, model Genesys 20, Waltham, MA, USA) set at 517 nm was used to measure the color reduction of DPPH. The AC was determined using the following formula:
where Asample is the absorbance of the sample in the presence of DPPH, and Acontrol is the absorbance of a solution that contains only DPPH solution.
The paper disk method, modified from Quyen and Quoc (2024), was used to measure antibacterial activity (AA). To begin, evenly spread 100 μL of bacterial suspension (0.5 McFarland standard concentration, corresponding to about 1.5×108 CFU/mL) onto Mueller-Hinton agar (MHA) medium with sterile paper disks. Sterile paper disks (6 mm in diameter) were inoculated with 5 μL of the oils, with gentamicin (10 μg/disk) and DMSO (5%, v/v) serving as positive and negative controls, respectively. The paper disks were incubated at 37°C for 24 h. The diameter of the inhibitory zone around the paper disk was measured to assess AA.
Statgraphics Centurion XVIII (Statgraphics Technologies, Inc., The Plains, VA, USA) was used for ANOVA and mean comparisons. A 95% confidence level (p<0.05) was calculated using the least significant difference (LSD) approach. Results are presented as mean±standard deviation (mean±SD). Principal component analysis (PCA) was conducted on the compound concentration data using covariance matrices to characterize the principal components of the three oil types and elucidate their compositional differences. The analysis was performed in R software (version 4.2.1; R Foundation for Statistical Computing, Vienna, Austria).
3. Results and discussion
According to the study’s color analysis results, the peanut oil samples (RPO, SPO, and BPO) showed significant color variation (Table 1). High L* (47.29) and low b* (22.12), representing light yellow and vivid hues, respectively, were observed in the RPO sample. This finding is consistent with a study by Salve and Arya (2018) in India, which likewise classified peanut oil as pale yellow and brilliant. In contrast, SPO and BPO showed far higher b* values (38.08 and 36.24, respectively) than RPO (22.12), indicating a deep/dark yellow hue. Visually, BPO appeared darker than SPO. The findings of Anyasor et al. (2009) in Nigeria, where peanut oil was characterized as amber and golden, are similar to this study.
The pressed oils from the three peanut varieties also exhibited differences in rheological properties, as seen by their kinematic viscosities, which ranged from 35.44 cP (RPO) to 38.85 cP (BPO). The viscosities of the peanut oil samples in this study tended to be comparable to or higher than those of soybean oil (24.5-15.0 cP), kulikuli oil (27.0-18.0 cP), and olive oil (29.0-17.0 cP), when compared to Davies (2016) study on the viscosities of some common seed oils in Nigeria. Interestingly, the viscosity of the BPO oil sample (38.85 cP) was comparable to that of crude palm oil (39.5 cP) and sunflower oil (39.5 cP), indicating similar possible molecular structure or molecular interactions between oil molecules. In addition, sample density did not change significantly (approximately 0.87 g/mL).
Table 2 shows that the BPO sample possesses the best yield (40.53%). Notably, our results were higher than those of Anyasor et al. (2009), who used the Soxhlet method to extract peanut oil in Nigeria (yield: 18.6-20.8%), and similar to those of Salve and Arya (2018), authors used the pressing method to isolate peanut oil in India (yield: 41.53-47%). This discrepancy suggests that variables beyond the extraction technique, such as peanut cultivar variation, seed maturity, and geographic environmental factors, may be crucial in influencing the oil recovery yield.
The acid value of RPO had the highest value (2.62 mg KOH/g). This is a crucial measure of oil quality, indicating a higher free fatty acids content than the SPO and BPO (approximately 0.94 mg KOH/g). Compared to previous studies, the AV of SPO and BPO were considerably lower than that of peanut oil in Kaduna, Nigeria (2.13-4.49 mg KOH/g) and in Mumbai, India (1.79-2.01 mg KOH/g) (Anyasor et al., 2009; Salve and Arya, 2018).
In addition, BPO exhibited the highest peroxide value (0.67 meq/kg), which was much lower than the values reported for peanut oils from India (5.58-13.05 meq/kg) but comparable to those observed in Nigeria (0.65-0.85 meq/kg) (Anyasor et al., 2009; Salve and Arya, 2018). This implies that the processing technique and storage circumstances affect the oil’s initial degree of oxidation. In comparison to studies conducted in Mumbai, India (191.67-368.38 mg KOH/g) and Aswan, India (189.41-194.75 mg KOH/g), the saponification value (SV) of the samples in this study (79.48-103.32 mg KOH/g) was much lower (Salve and Arya, 2018, Zahran and Tawfeuk, 2019). Notably, the RPO’s ester values (100.69 mg KOH/g) were significantly higher than those of the SPO and BPO samples (78.55-80.89 mg KOH/g). This may be related to the changes in triglyceride composition during processing and is consistent with the declining trend in the saponification value.
These findings suggest that the processing technique and the source of the raw material significantly affect the chemical characteristics of peanut oil. While the SPO sample demonstrated greater stability in quality metrics, the BPO sample outperformed the others in terms of extraction efficiency but should be cautious about the oxidation index. The distinction from earlier research also underscores the importance of setting appropriate quality standards for each distinct geographic location.
Table 3 shows that the fatty acid composition of the three peanut varieties differed significantly. The five main fatty acids that were found to contribute to the distinctive qualities of peanut oil were palmitic acid (C16:0), stearic acid (C18:0), arachidic acid (C20:0), oleic acid (C18:1), and linoleic acid (C18:2).
The RPO has notable for having the highest percentage of linoleic acid (40.56%) among the PUFA group, a necessary omega-6 fatty acid. In contrast, BPO showed a higher overall MUFA content (59.06%). Notably, SPO exhibited a high oleic acid concentration (47.33%), comparable to that of olive oil, which is well-known for its cardiovascular advantages (Bermudez et al., 2011).
One noteworthy feature of the BPO type is its low SFA level (7.92%), which is perfect for diet products. The composition of fatty acids differs significantly when compared to previous studies. In contrast to this study’s palmitic acid content (11.08-13.20%), an Indian study found a higher amount of oleic acid (50.21%) but a lower proportion of linoleic acid (28.46%) (Salve and Arya, 2018).
Interestingly enough, 17-octadecenoic acid (23.18%) was specific to BPO. This fatty acid was also detected in peanut oil in the study of Qiu and Zhang (2021). In addition, it also accounts for a small proportion (3%) of peanut oil from Saudi Arabia, where the total unsaturated fatty acid content was dominated by oleic and linoleic acids (Al-Rajhi et al., 2025). 17-octadecenoic acid is a unique compound recognized for its potential to exhibit diverse biological activities, including antibacterial and anti-inflammatory properties. Furthermore, its distinct triple bond structure makes it a versatile chemical precursor for the synthesis of complex organic compounds (Younis-Khalaf et al., 2021). This highlights a distinctive compositional feature of BPO compared to groundnut oils from other regions, suggesting potential differences in biological functionality and application. This points out that different varieties may have produced distinct metabolic pathways or secondary metabolites (Al-kashef et al., 2024). Similarly, traces of gondoic acid and petroselinic acid were found in SPO, indicating distinct metabolic processes (Teoh and Ng, 2013).
The oleic/linoleic acid ratios of SPO (1.43), RPO (1.07), and BPO (1.09) showed nutritional promise although falling short of the optimal range (1.67-3.0) for frying oil, as suggested by Issariyakul et al. (2008). As an alternative to hydrogenated oils, combining oils from these types with other oils with better fatty acid ratios can help maximize oil quality for frying. This will meet technical requirements for healthy frying oils and improve nutritional value.
Fatty acid composition analysis by GC-MS, combined with principal component analysis (PCA), revealed significant differences in chemical composition among the three peanut varieties (RPO, SPO, and BPO) (Fig. 1). The samples’ distinct separation on the PCA plot showed that the varieties’ fatty acid composition varied consistently. The first principal component (PC1) accounted for 68.96% of the total variance, while the second principal component (PC2) accounted for 31.04%.
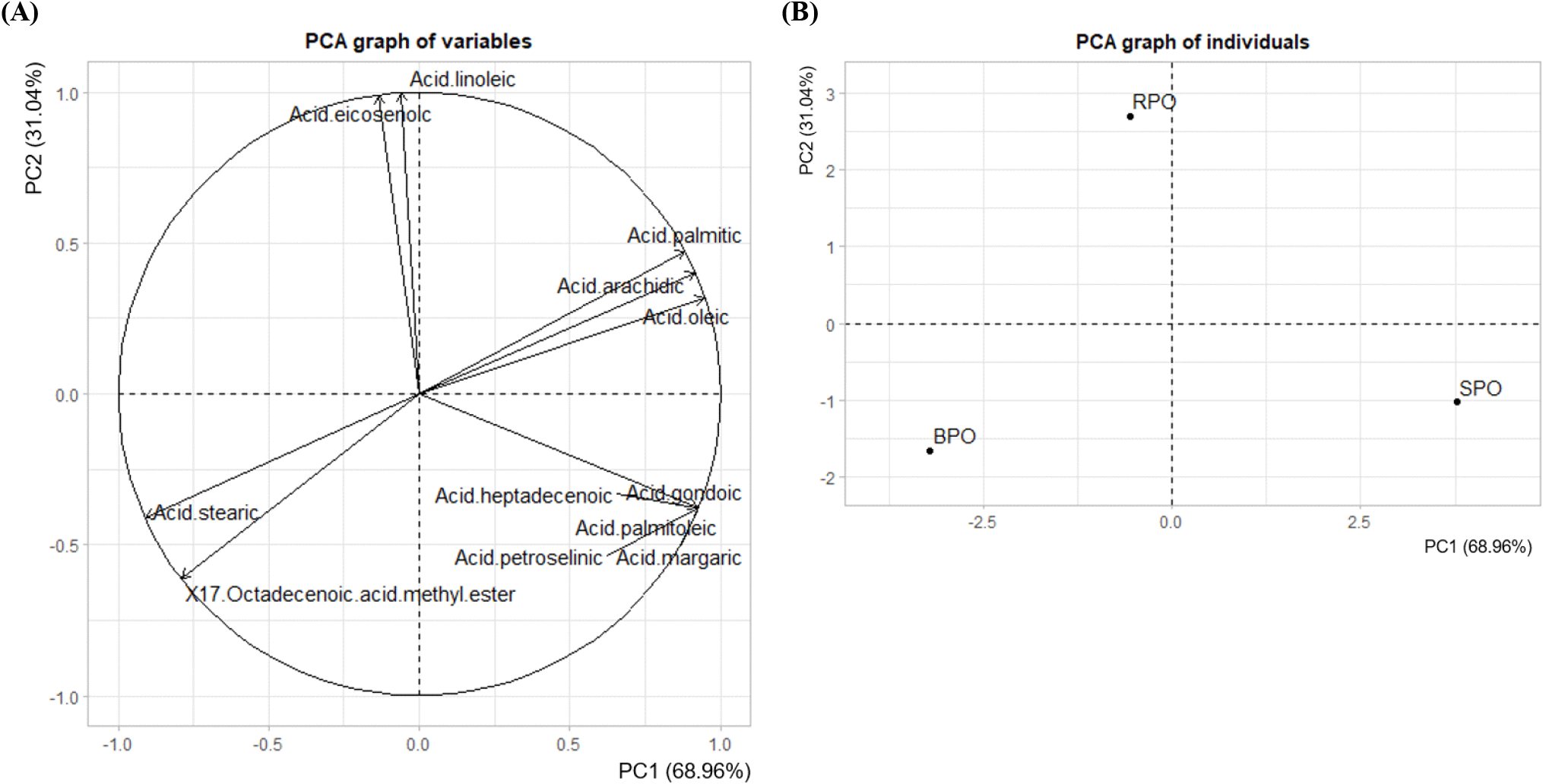
RPO had a high linoleic acid content (40.56%) and was primarily differentiated from SPO and BPO along PC1. BPO, on the other hand, was distinguished by its high content of 17-octadecenoic acid (23.18%), while SPO was separated along PC2 due to its elevated oleic acid content (47.33%). These three fatty acids strongly influenced the direction and magnitude of the vectors on the variable plot (Fig. 1A), indicating that they played key roles in driving the separation among the cultivars. The PCA biplot and clustering (Fig. 1B) clearly reflected the chemical diversity and distinct fatty acid profiles among peanut oil samples, highlighting potential implications for nutritional quality and functional properties.
The results showed that all three varieties had antioxidant capacity (AC), with the increasing order being SPO (14.51%), BPO (19.35%) and RPO (23.03%) (Table 4). The RPO sample with the greatest linoleic acid (PUFA) content (40.61%) also had the strongest AC. This implies that the oil’s potent AC may be related to its high linoleic acid content. Farhoosh (2015) reported a possible correlation between linoleic acid concentration and antioxidant activity in vegetable oils, suggesting that certain unsaturated fatty acids might contribute to oxidative stability. Additionally, Lalithadevi et al. (2018) emphasized that fatty acid composition can influence antioxidant mechanisms, opening the door to future investigations into their specific roles and applications. While the current study did not directly evaluate this relationship, the observed trends support the hypothesis that fatty acids, such as linoleic acid, modulate antioxidant behavior. The AC of peanut oil was 14.51-23.03% in our study, which is much lower than the results of studies conducted in Taiwan (42.02-52.34%) and India (30.37-39.34%) (Ciou et al., 2021; Taha et al., 2012). Different pressing temperatures and extraction techniques (cold pressing, hot pressing, or organic solvents extraction) can account for this discrepancy.
| RPO1) | SPO | BPO | |
|---|---|---|---|
| DPPHRSA (%) | 23.03±2.432)c3) | 14.51±2.06a | 19.35±0.34b |
This study assessed the antibacterial activity (AA) of peanut oils against two Gram-negative bacterial strains, S. Enteritidis and E. coli, as shown in Table 5. The results showed that the positive control (Gentamicin) exhibited strong AA against both E. coli (19.88 mm) and S. Enteritidis (24.05 mm).
According to the study’s findings, neither RPO, SPO, nor BPO exhibited any inhibitory effect on the growth of the two tested Gram-negative bacteria. This aligns with earlier research findings. According to Sutrisno et al. (2021), peanut oil exhibited no discernible antibacterial activity against either Gram-positive or Gram-negative bacteria (S. aureus and E. coli). Similarities with other research further corroborate the findings of this study. This may be attributed to the oils’ high content of long-chain unsaturated fatty acids such as oleic and linoleic acids, which generally have weak or negligible antimicrobial effects. Moreover, the low levels of medium-chain saturated fatty acids and the absence of phenolic compounds or free fatty acids further limit their antibacterial potential. According to Sutrisno et al. (2021), the esterification of fatty acids into methyl esters significantly reduces or eliminates antibacterial activity. Since peanut oil consists mainly of triglycerides rather than free fatty acids or potassium salts, its structural form is not conducive to disrupting microbial membranes. Furthermore, its high non-polarity and molecular structure hinder interactions with the bacterial cell envelope, particularly in Gram-negative bacteria, which possess an additional outer membrane that acts as a protective barrier.
4. Conclusions
This study successfully identified and evaluated the fatty acid composition and antioxidant activity of pressed oils from three different peanut varieties. The significant differences in the proportions of major fatty acid groups (SFA, MUFA, and PUFA) and antioxidant activity among varieties demonstrated each variety’s genetic diversity and application potential. RPOs were known for their high PUFA content and antioxidant activity, whereas SPOs were high in SFA and BPOs had a high MUFA level. These findings provide an important scientific foundation for selecting the best peanut variety for vegetable oil production, aim to produce high-nutritional-value products with improved stability. Simultaneously, the study adds to a better knowledge of the potential of local peanuts.










