1. Introduction
Cashew nuts (Anacardium occidentale), known for their creamy texture and rich taste, undergo critical processing stages before reaching consumers, with roasting being a pivotal step. This technique significantly impacts the kernel’s physical and theoretical properties, including color, texture, size, oil content, protein quality, and antioxidant activity. A comprehensive understanding of the complex interaction between roasting settings and cashew kernel attributes is essential for enhancing quality, marketability, and consumer appeal (Chen et al., 2024).
The cashew nuts are the raw nuts in their shells, while the cashew kernels are the edible kernels derived from the nuts. Cashew nut is the edible seed of the cashew tree that is renowned for its rich taste and nutritional content. Cashews are a lucrative agricultural product that enhances the economies of many cashew-producing areas. The cashew kernel, housed within a hard shell beneath the cashew apple, undergoes multiple processing operations before reaching consumers, including harvesting, shelling, drying, grading, and packaging. Among these, roasting is a crucial process that significantly improves cashews’ flavor profile and overall quality. Sanchez et al. (2024) stressed the necessity of roasting to get the optimum color in cashew kernels. The Maillard reaction, initiated by heat during roasting, leads to browning and caramelization, generating appealing color variations, ranging from light golden to deep brown. These hue variants respond to varied customer tastes, impacting purchase choices. Notably, roasting characteristics like temperature and exposure time strongly influence the final color, necessitating fine management to obtain specified hues.
Roasting significantly changes the final product’s sensory qualities, nutritional value, and shelf life. Chen et al. (2024) emphasize the necessity of roasting for generating the appropriate color, texture, and taste, which reveals the real taste potential of cashew kernels. Olatidoye (2021) described the intricate interaction of chemical processes during roasting, yielding volatile molecules responsible for unique fragrance and taste qualities.
While the role of roasting for cashew quality is universally acknowledged, the optimal roasting settings for reaching the essential balance of physical and theoretical attributes are still contested (Animasahun et al., 2024; Liu et al., 2023). Existing research delivers helpful information, but it typically concentrates on single effects of temperature or time, neglecting their combined influence and the delicate relationship with distinct cashew varieties and origins. Furthermore, there are inconsistencies in technique and reporting, making it difficult to establish definite judgments. This study intends to solve these constraints by carefully examining the individual and combined effects of roasting temperature and duration on a variety of physical and theoretical features of cashew kernels derived from cashew tree (Anacardium occidentale).
2. Materials and methods
The semi-processed cashew kernels used for the research were obtained from the Cocoa Research Institute of Nigeria, Ibadan, Oyo State, Nigeria. They were pre-packaged in self-standing Ziplock pouches and transported to the laboratory for further processing and analysis.
The roasting of the cashew nut kernel was carried out following the method described by Liaotrakoon et al. (2016). The cashew kernels were roasted using a dry air oven (Genlab, London, UK) at temperatures of 120°C, 130°C, 140°C, and 150°C for 50 min, followed by cooling to room temperature, and stored in Ziplock pouches for further processing. The roasting processes were done in triplicate. The roasted kernels were then milled using a Bajaj grinder (GX-3701, New Delhi, India). Powder was stored in Ziplock storage bags until further analysis.
The extraction of oil from the cashew nut was done using the method described by Awolu et al. (2013). Finely ground cashew kernels (100 g) were enclosed in a muslin cloth, placed within a thimble, and positioned inside a Soxhlet extractor. Exactly 1,000 mL of n-hexane (99.9%) was measured into a round-bottom flask and heated with a heating mantle at 60°C for 6 h. The extracted oil was recovered by distillation of n-hexane. Extracted oil samples were labelled as CO120, CO130, CO140, and CO150, corresponding to their respective roasting temperatures.
The quantity of oil obtained from a given mass of cashew kernels is defined as oil yield and can be mathematically represented according to Equation (1).
Where the weight of oil extracted is the total mass of oil obtained from the extraction process, and the weight of raw materials used is the mass of the raw materials (cashew kernel) used in the extraction process.
A HunterLab colorimeter (HunterLab, MI, USA) with an optical sensor was used to determine the color parameters of cashew nut oil, which include L* (lightness, ranging from 0 for black to 100 for white), a* (with negative values indicating green and positive values indicating red), and b* (where negative values signify blue and positive values signify yellow). The instrument was calibrated to L*=96.9, a*=−0.04, and b*=1.84 before measurements were taken. Several color indices, such as total color difference (ΔE), degree of whiteness (%W), browning index and yellowness index, were computed from the results following the method outlined by Olawoye and Gbadamosi (2020).
About 2 g of the sample was weighed into a dry 250 mL Erlenmeyer flask. To dissolve the oil, 30 mL of a solvent mixture containing equal portions of 95% ethanol and diethyl ether was added. The resulting solution was titrated against 0.1 N methanolic potassium hydroxide, with 0.5 mL of phenolphthalein indicator added. Titration continued until a faint pink color remained for 15 sec. A blank titration, conducted under identical conditions but without sample addition, was also performed. Equation (6) was used to determine the acid value (Awolu et al., 2013).
Where:
V = volume of KOH solution used in titration
N = Normality of the KOH solution
W = Weight of the sample (g)
56.1 = Molecular weight of KOH
Fourier transform infrared (FT-IR) spectroscopy was employed to analyze the functional groups in cashew kernel oil samples. The FT-IR spectrometer scanned each sample 32 times with a resolution of 4cm−1, using an infrared light source that covered a wavenumber range of 4,000 to 400 cm−1. The generated infrared spectrum was Fourier transformed and recorded in absorbance mode, producing an interferogram representing wavenumber versus absorption. Spectrum acquisition was performed using the IR solution software. Following the approach outlined by Coates and Sanders (2000), functional groups were identified by comparing the vibration frequencies of wavenumber from the sample’s FT-IR spectrograph (Olawoye et al., 2022).
A 2.5 g portion of the oil sample was placed in a 250 mL conical flask, followed by the addition of 25 mL of ethanol. The mixture was heated until it reached its boiling point, then allowed to cool. After cooling, 2-3 drops of phenolphthalein indicator were added. The sample was titrated against a standardized 0.1 M KOH solution with continuous vigorous shaking until a persistent pink color signaled the endpoint (Onwuzuruike et al., 2024). The free fatty acid content was then determined using the equation below.
Where 28.2 is constant and,
V = volume in millilitres of standard potassium hydroxide used
C = concentration of potassium hydroxide solution
W = weight in grams of a sample
A titrimetric approach was employed to determine the saponification value of the oil (Onwuzuruike et al., 2024). A sample of 2 g was precisely measured and transferred into a 250 mL conical flask, where it was combined with 25 mL of 10% alcoholic potassium hydroxide (KOH). The mixture was then subjected to heating for 30 min with periodic shaking. Following cooling, 0.5 mL of a 1% phenolphthalein solution was added. A blank titration without the oil sample was performed under identical conditions. The saponification value was then obtained using the equation below.
Where:
B = volume (mL) of HCl used in the blank titration
S = volume (mL) of HCl used in the oil sample titration
N = normality of HCl
W = weight of the oil sample (g)
To determine the iodine value, a traditional titration method was employed (Olagbaju et al., 2023). The conventional procedure utilizes chloroform or carbon tetrachloride as solvents, both of which pose potential hazards. The oil sample was dissolved in the selected solvent, and a fixed quantity of iodine solution was added. Following a reaction time of approximately 30 min, the residual iodine was titrated against a sodium thiosulfate solution, thereby indirectly quantifying the iodine uptake. The iodine value was obtained using the following Equation (9);
Where:
S = volume (mL) of sodium thiosulfate solution for back-titration of the sample (mL)
B = volume (mL) of sodium thiosulfate solution for back-titration of the blank (mL)
N = normality of the thiosulfate solution
W = weight of the sample (g)
Following the AOAC Official Method (AOAC, 2005), the p-AV analysis was performed. An appropriate aliquot of oil sample (0.5 g) was precisely weighed and introduced into a 25 mL graduated flask, with hexane added to reach the calibration line. The absorbance of the test tube containing only hexane was recorded at 350 nm (A1). Following this, 5 mL of the oil sample (m) was transferred into a separate test tube, and 1 mL of p-AV solution (0.25 g/100 mL glacial acetic acid) was added. After allowing the reaction to proceed for 10 min, the absorbance (A2) was measured at 350 nm, using a reference solution prepared with hexane and p-AV without the oil sample. The p-AV value was subsequently derived using the equation below.
Where:
W = sample weight (g)
A 0.4 g oil sample was introduced into a screw-capped test tube, followed by the addition of three drops of antioxidant solution (Tesfaye et al., 2018). The solvent was evaporated under reduced pressure using a rotary evaporator at a water-bath temperature of 35-40°C. Subsequently, 3 mL of TBA solution and 17 mL of trichloroacetic acid (TCA) solution were added. After flushing the test tube with nitrogen (N2) gas, it was immediately sealed and heated in a boiling water bath at 100°C for 30 min until the characteristic color developed. After cooling to room temperature, 5 mL of chloroform was added and briefly mixed using a vortex mixer. A 15 mL portion of the solution was transferred into a glass centrifuge tube and centrifuged at 3,000 rpm for 10 min. If the aqueous layer remained unclear, another centrifugation was performed at 10,000 rpm for an additional 10 min. A small portion of the clarified aqueous layer was transferred, and its absorbance was measured at 532 nm. A blank test, following the same procedure without the oil sample, was conducted as a control. The TBA value was then calculated using Equation (11).
Where:
Abs = absorbance at 532 nm
W = weight of fat in volume of extract (g)
F = factor = 46
All data were generated in triplicate. Statistical analysis was performed to evaluate the collected data, which were expressed as means±standard deviations. A one-way analysis of variance (ANOVA) was employed, and Duncan’s Multiple Range Test (DMRT) was used to determine significant differences among treatment means at p<0.05. The statistical methodology was applied following the approach of Steel, with calculations conducted using XLSTAT version 2019 (Addinsoft Inc., USA).
3. Results and discussion
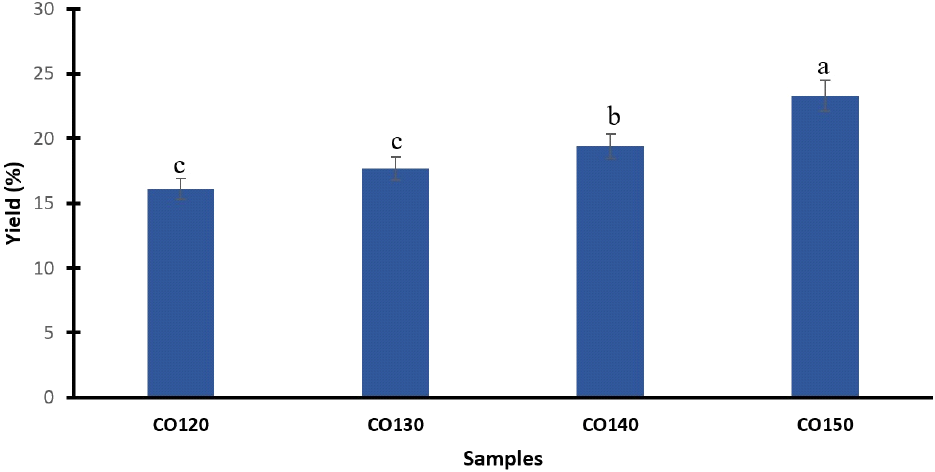
Table 1 and Fig. 1 present the percentage yield of the cashew kernel oil extracted using the Soxhlet extraction method. The table shows oil yields for cashew kernels roasted at different temperatures (120-150°C) while maintaining a constant roasting duration of 50 min. The results indicate that the oil extraction yield was optimized under specific conditions. Notably, kernels roasted at 150°C for 50 min yielded the highest oil yield (23.3%), followed by those roasted at 140°C (19.4%) and 130°C (17.7%), while the lowest yield (16.1%) was observed at 120°C. This study recorded an oil yield higher than the 11.87% previously documented by Idah et al. (2014). The findings suggest that increasing the roasting temperature enhances the extraction efficiency of oil from cashew kernels. The increasing trend in oil yield with temperature indicates that higher roasting temperatures facilitate better oil extraction, likely due to changes in the physical and chemical structures of the kernels that make the oil more accessible. These results imply that, for commercial oil production, roasting cashew kernels at higher temperatures may be advantageous to maximize oil yields.
| Samples1) | Processing conditions | Yield (%) | |
|---|---|---|---|
| Roasting temperature (°C) | Roasting time (mins) | ||
| CO120 | 120 | 50 | 16.1±0.862)c3) |
| CO130 | 130 | 50 | 17.7±0.61c |
| CO140 | 140 | 50 | 19.4±0.90b |
| CO150 | 150 | 50 | 23.3±1.07a |
The color characteristics of cashew kernel oil at different roasting temperatures are presented in Table 2, with Fig. 1 illustrating the correlation between these color parameters. The roasting process significantly affected the color of cashew kernels, with higher temperatures resulting in darker kernels. This color change is indicative of Maillard reactions occurring during roasting (Olawoye et al., 2024).
1) CO120, cashew kernel oil roasted at 120°C; CO130, cashew kernel oil roasted at 130°C; CO140, cashew kernel oil roasted at 140°C; CO150, cashew kernel oil roasted at 150°C.
Table 2 presents various color parameters for cashew kernel oil at different roasting temperatures (120°C, 130°C, 140°C, and 150°C). Color parameters measured in this study include L* (lightness), a* (red-green), b* (yellow-blue), chroma (C*), hue angle (H), whiteness index (WI), yellowness index (YI), and blue index (BI). Statistical analysis indicated a significant difference (p<0.05) in the L* values of the oil. The highest lightness value is observed at 130°C (30.90), indicating that this oil is the lightest in color. In contrast, the oil roasted at 150°C has the lowest lightness (27.92), suggesting a darker color due to increased roasting. In studies related to sunflower oil, authors noted that higher lightness values corresponded to refined oils, where pigments and impurities had been removed (Maskan, 2003). This could indicate that CO130 contains fewer pigments than CO150. The a* values remained relatively stable across all samples (p>0.05), ranging from 2.58 to 2.66, suggesting that roasting temperature did not significantly influence the red-green balance. Similar trends have been reported in olive oil (Caponio et al., 2013). This uniformity could be due to the natural content of carotenoids in cashew oil, which imparts a stable red tint across all samples. This indicates that roasting temperature does not significantly affect the red-green balance in the oils. In contrast, significant differences (p<0.05) were observed in b* values, which represent the yellow-blue axis. The highest b* value was recorded in CO130 (11.01), indicating more yellow tones, while CO140 had the lowest (9.81). This variation in yellowness may be attributed to differences in carotenoid concentration or oxidation levels, similar to findings in palm oil, where yellowness is a key indicator of carotenoid content (Silva et al., 2010). Lower b* values in CO140 suggest lower carotenoid content or partial degradation due to oxidation. This suggests that moderate roasting enhances the yellow hue. The whiteness index (WI) follows a similar trend to L*, with CO130 showing the highest whiteness (29.18) and CO150 the lowest (27.18). Supporting the observation that higher roasting temperatures lead to darker oils. Oils with higher whiteness values are often considered to have undergone more efficient refining processes, removing impurities that could darken the oil. This finding aligns with studies on palm oil by Rossi et al. (2001), where refined oils exhibited higher whiteness indices.
The ΔE values indicate significant perceptual color differences (p<0.05) among the samples, with CO130 showing the largest color difference (32.15) and CO150 the smallest (29.78). Differences in ΔE may result from different processing methods, such as cold pressing versus heat treatment, as previously documented in studies on sesame and flaxseed oils (Ifa et al., 2021). Larger ΔE values typically indicate more processing or degradation of natural pigments.
The yellowing index (YI) results suggest that CO120 and CO130 exhibited more pronounced yellow coloration, indicating higher carotenoid content or lower levels of degradation. Conversely, CO140 displayed the lowest yellowness and blueness values, suggesting it may have undergone more oxidation or processing. These findings are consistent with prior research on palm oil, where YI is frequently used to monitor oil degradation during storage (Singkhonrat et al., 2019). Higher YI values typically indicate higher concentration of natural pigments or reduced oxidation, while lower values suggest degradation.
Table 3 and Fig. 2 present the chemical composition of the extracted cashew kernel oil at different roasting temperatures (120°C, 130°C, 140°C, and 150°C) for a constant duration of 50 minutes. The measured properties include free fatty acid value, saponification value, acid value, iodine value, p-anisidine value, and thiobarbituric acid value.
1) CO120, cashew kernel oil roasted at 120°C; CO130, cashew kernel oil roasted at 130°C; CO140, cashew kernel oil roasted at 140°C; CO150, cashew kernel oil roasted at 150°C.
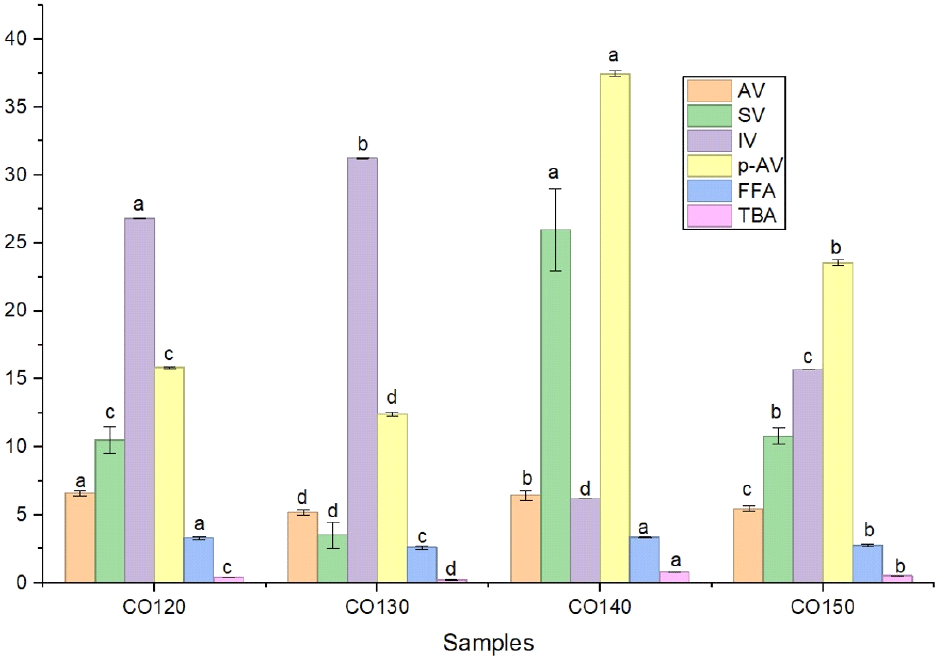
The acid value indicates the presence of free fatty acid content in the oil, typically increasing as a result of oxidation and hydrolysis. CO140 showed a marginally higher acid value (6.45 mg KOH/g) compared to CO130. Suggesting that higher roasting temperatures accelerate the breakdown of triglycerides, releasing more free fatty acids. Cashew kernel oil exhibited the following acid values: CO120 (6.59), CO130 (5.19), CO140 (6.45), and CO150 (5.47) mg KOH/g. Compared to previous studies, the acid values obtained in this study are higher than those reported by Nkwocha et al. (2016) (3.41±0.07 mg KOH/g) and Aremu et al. (2006) (0.82 mg KOH/g) but lower than the 15.7 mg KOH/g recorded by Akinhanmi et al. (2008). The relatively low acid values confirm that the oil is in a stable, non-degraded state and is suitable for applications in paint and varnish production. The lowest acid value is observed at 130°C, indicating better oil quality with less free fatty acid content at this temperature.
The saponification value (SV), an indicator of the average molecular weight of fatty acids in cashew kernel oil, was found to be 10.51±0.98, 3.51±0.99, 25.97±3.02, and 10.81±0.58 mg KOH/g. The low saponification value of oil extracted from cashew nut roasted at 130 could be due to the fact that either the oil contains a high proportion of long-chain fatty acids, which are difficult to saponify, or that continuous heating causes the degradation of the oil thereby increasing the proportion of shorter-chain free fatty acids and triglycerides that are easier to saponify and potentially making the oil less suitable for certain application. The saponification values recorded in this study are notably lower than the 187 mg KOH/g reported for groundnut oil (Atasie et al., 2009). This lower value indicates that cashew kernel oil may not possess the required properties for effective soap formulation. Saponification values vary significantly, with the highest value at 140°C (25.97) and the lowest at 130°C (3.51). A higher saponification value suggests a greater amount of ester bonds in the cashew kernel oil, which signifies higher oil quality. Higher SVs typically suggest shorter-chain fatty acids, whereas lower SVs suggest longer-chain ones. The high value in CO140 indicates the presence of shorter-chain fatty acids or increased breakdown due to higher roasting temperatures. This aligns with studies on other nuts, which show that heating affects fatty acid profiles and can increase SVs due to chain scission (Boateng et al., 2016).
The iodine value quantifies the degree of unsaturation in organic compounds, serving as an indicator of double bond reactivity. The iodine values show a notable decrease from 26.80 at 120°C to 6.20 g I2/100 g at 140°C. The high iodine of CO120 indicates a high degree of unsaturation in the oil, meaning more double bonds in the fatty acid chains, which leads to increased susceptibility to oxidation, a shorter shelf life, and lower stability. A decreasing iodine value with increasing roasting temperature suggests oxidative degradation of double bonds (Onyeike and Acheru, 2002), which aligns with the CO140’s results under higher temperature conditions. The low iodine value indicates a lower level of unsaturation and classifies the cashew kernel oil as a non-drying oil. This is in alignment with recorded values for most edible oils, typically ranging between 80 and 100 g/100 g (Atasie et al., 2009).
The p-Anisidine values (PAV) reflect the presence of secondary oxidation products, particularly aldehydes, which form during the later stages of lipid oxidation. CO140 shows the highest PAV (37.45), followed by CO150 (23.55), CO120 (15.85), and CO130 (12.40), indicating that prolonged exposure to higher temperatures significantly increases oxidation in the cashew kernel oil. The elevated PAV in CO140 and CO150 suggests that higher roasting temperatures facilitate secondary oxidation, leading to more aldehyde formation. Studies on thermal oxidation in oils corroborate this, showing that increased heat accelerates oxidative rancidity. The highest PAV is recorded at 140°C (37.45°C), suggesting that this temperature may lead to increased oxidation products in the cashew kernel oil. This is lower than the value 50.01±3.17 reported by Ali et al. (2013) in sunflower oil.
The FFA is higher at lower temperatures (CO120) and decreases as roasting temperature increases, which might indicate more stable oils at higher temperatures due to reduced free fatty acid formation. Free fatty acid value signifies the amount of free fatty acids due to triglyceride hydrolysis. CO140 has the highest FFA (3.36%), while CO130 has the lowest (2.60%), which may result from the breakdown of triglycerides at higher roasting temperatures. Studies confirm that higher temperatures contribute to triglyceride hydrolysis, increasing FFA in oils (Liao et al., 2019). The pattern in this study supports the effect of roasting on fatty acid liberation. The free fatty acid content of cashew kernel oil was determined to be 3.30%, 2.60%, 3.36%, and 2.74%, respectively. These values are significantly lower than the 36.09±1.12% reported by Liao et al. (2019), suggesting that the oil, if refined, could be suitable for consumption. Additionally, the low free fatty acid content implies a reduced tendency for oxidative deterioration, which can lead to off-flavor formation, as noted by Abitogu and Borokini (2009).
The Thiobarbituric acid value, which measures malondialdehyde (MDA), a byproduct of lipid peroxidation. CO140 has the highest TBA value (0.79), indicating greater oxidative deterioration, while CO130 has the lowest (0.21). This agrees with studies on lipid oxidation, where elevated temperatures accelerate peroxidation, raising TBA levels (Choe and Min, 2006). The Thiobarbituric acid value measures lipid oxidation; it is lowest at 130°C (0.21) and highest at 140°C (0.79), indicating that higher roasting temperatures may lead to increased oxidation levels in the cashew kernel oil. Similar results were obtained by Silva et al. (2010) and Pal et al. (2015). There were notable differences (p<0.05) between the TBA values of the cashew kernel oil samples.
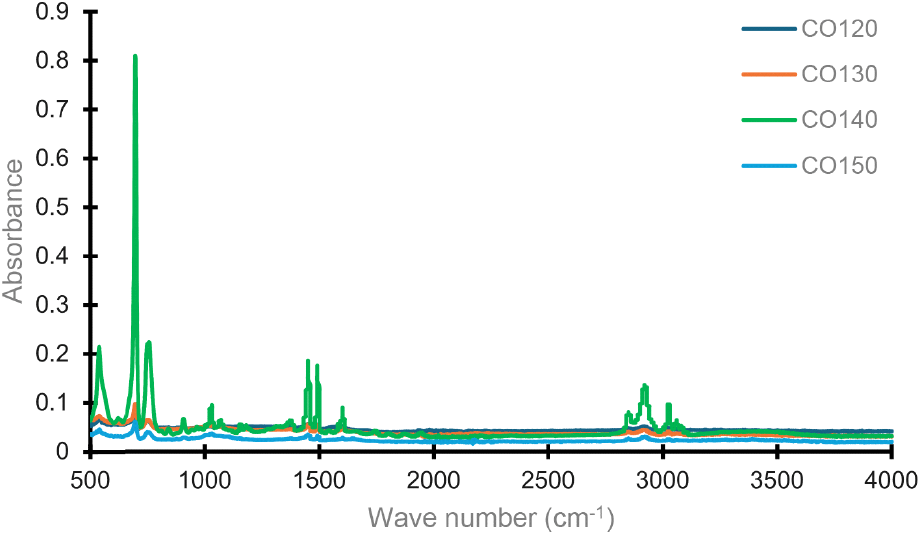
Fig. 3 shows the characteristic FT-IR spectra of the cashew oil as affected by processing temperature. The peaks at 2,920 cm−1 and 2,850 cm−1 relate to C-H stretching, corresponding to the asymmetric and symmetric stretching of CH2 groups, which are part of the aliphatic chains of fatty acids. A strong absorption band at 1,745 cm−1 (C=O stretching) indicates the presence of ester carbonyl groups, typical of triglycerides, which are the major components of oils. The bands at 1,460-1,370 cm−1 (C-H bending) region emerge from CH2 and CH3 bending vibrations, indicating the aliphatic character of the fatty acid chains in the oil. The region lies between 1,230-1,160 cm−1 (C-O stretching): This region exhibits peaks due to the stretching of ester bonds, typically for triglycerides in oils, while the region at 720 cm−1 (CH2 rocking): A peak here shows long-chain aliphatic hydrocarbons, a common characteristic in oils containing fatty acids. This FTIR spectra indicate the presence of fatty acids and triglycerides, which are important components of cashew kernel oil. The increase in roasting temperature caused bands to shift to higher frequencies (blueshift), an increase in intensity, and to become narrower as molecules become more ordered and interatomic distances shorten. The spectrum is similar to that of normal vegetable oils. Cashew oil extracted at 140°C resulted in higher intensity compared to other processing temperatures.
4. Conclusions
In conclusion, the optimal conditions were identified as roasting at 130°C for 50 min, which balanced desirable physical properties with maximum oil yield while preservingthe quality of the oil owing to its reduced acid value, p-anisidine value, TBA value, saponification, as well as free fatty acid value.The study showed that the percentage oil yield and rate of color changes were significantly affected by temperature and time over the temperature range used in this study. The effect of roasting temperature and time was found to be more significant on the samples roasted at higher temperatures (especially 140°C) appear to promote oxidation, which could negatively impact the flavor and nutritional quality of the oil. The study showed that increase in roasting temperature caused bands to shift to higher frequencies (blueshift), an increase in intensity, and become narrower as molecules become more ordered and interatomic distances shorten. Overall, this study suggests potential modification ofthe quality of cashew kernel oil by simply optimizing the time and temperature profiles during roasting.










