1. Introduction
Skipjack tuna (Katsuwonus pelamis) is a major component of Philippine fisheries, accounting for 70% of canned tuna exports valued at more than PHP 50 billion annually and serving as a primary dietary protein source (Delfino, 2023). This highly migratory pelagic species is commonly infected with acanthocephalan helminths, characterized by a retractable proboscis armed with hooks that attach to the intestinal mucosa, causing mechanical damage, inflammation, ulceration, and potential perforation (Beaver, 1986; Perrot-Minnot et al., 2023).
Parasitological surveys in Philippine waters have identified Rhadinorhynchus sp. as highly prevalent in skipjack tuna intestines, with infection rates reaching 100% and mean intensities exceeding 50 worms per heavily infected fish in certain cohorts (Briones, 2015; Dwi Pramardika et al., 2024). Although predominantly intraluminal, high parasite loads are associated with intestinal ulceration, hemorrhage, and perforation, as documented in affected tunas (Pambudi et al., 2021; Shibata et al., 2014; Shibata et al., 2020). However, no studies have reported extraintestinal migration to the hypaxial and epaxial musculature, which are the principal edible fillet portions processed for raw, sashimi, poke, or undercooked consumption worldwide (Al-Zubaidy et al., 2022; Nguyen et al., 2021).
Extraintestinal acanthocephalan localization in the musculature, although undocumented in Philippine skipjack tuna, represents a critical food safety and quality hazard. Encysted parasites evade routine visual candling inspections, forming detectable nodules or cysts during filleting that fail Hazard Analysis and Critical Control Points (HACCP) verification, trigger noncompliance with Codex Alimentarius and FDA parasite-free standards, and necessitate product rejection or recall (Piras et al., 2020). Muscle contamination induces sensory defects, including off-flavors, textural degradation, discoloration, and reduced shelf life, compromising marketability despite the absence of documented zoonotic transmission from fish acanthocephalans to humans. Comparative evidence from other hosts supports the feasibility of this migration pattern. Experimental Centrorhynchus spp. infections in rodents result in gut perforation and hypaxial muscle encystment under heavy burdens, whereas natural Profilicollis spp. infections in sea otters demonstrate intestinal breach and tissue invasion (Grewelle et al., 2023; Mayer et al., 2003).
In Mati City, Davao Oriental, a major landing site processing approximately 5,000 tons of skipjack annually, the complete lack of extraintestinal infection data impedes risk-based assessments under Bureau of Fisheries and Aquatic Resources (BFAR) protocols, leaves export products vulnerable to quality downgrades, and undermines compliance with international seafood safety benchmarks.
This study examined 96 skipjack tuna from Mati City using gross inspection, artificial pepsin digestion, and microscopic morphological analysis to determine the prevalence, mean abundance, and intensity of intestinal and extraintestinal acanthocephalan infections in the fish. The findings were evaluated through site-assurance-density-epidemiology (SADE) risk categorization to establish detection thresholds, validate inspection efficacy, and recommend targeted interventions, including enhanced freezing, depuration standards, and surveillance protocols, to ensure food safety, product quality, and regulatory compliance in the Philippine tuna industry.
2. Materials and methods
A total of 96 skipjack tuna samples were systematically collected from the Mati City Fish Port in the Philippines over a two weeks period in April 2023, which corresponds to the hot, dry season in the country (Fig. 1). Sampling was conducted daily during the early morning hours (4:00-6:00 a.m.) to ensure the freshness and representativeness of the catch obtained from local fishing vessels.
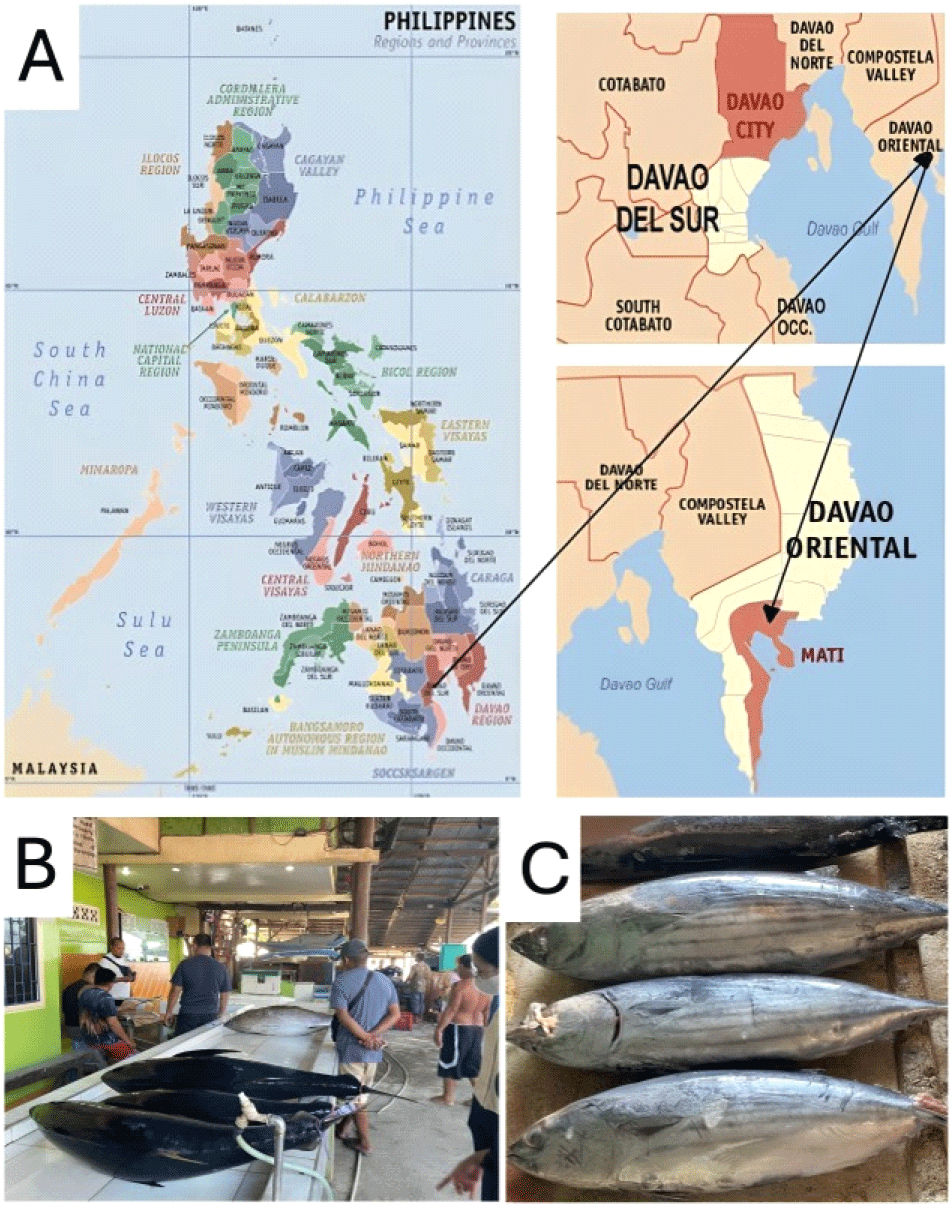
Skipjack tuna samples were collected and selected based on established criteria: fish samples had to be collected fresh from the port (not frozen) and within the commercial fork length (FL) range of 28-52 cm (Bintoro et al., 2021; Satria et al., 2021). During each sampling, two batches of fish were collected at the port. The first 16 skipjack tuna that met the criteria were selected from the first batch, and the remaining 16 from the second batch, completing 32 skipjack tuna samples per sampling event. Fish were collected systematically rather than randomly, following consistent selection criteria to ensure the representativeness of the local commercial catch.
Each fish was weighed to the nearest kilogram (0.93±0.16 kg), and the total length (38.92±6.00 cm) was measured as the fork length (i.e., from the tip of the snout to the longest caudal fin). The fish samples represented fresh, commercially sized skipjack tuna typically landed at the port, excluding unusually small and large individuals. Between the two weeks allocated for sample collection and examination, sampling was conducted three times, with 32 fish collected per event to reach the desired sample size of 96 specimens.
Post-mortem migration of parasites may occur after catching fish or during the decay period, which is a well-recognized phenomenon observed by parasitologists and fishers. Therefore, the collected fresh fish were placed in plastic bags containing water with ice (slushed and broken pieces) inside a styrofoam box to reduce spoilage growth and maintain the fish flesh under conditions as close as possible to those of fresh fish during transport to the laboratory.
The minimum requirements set by the Bureau of Fisheries and Aquatic Resources (BFAR) of the Philippines require the collection of at least ten moribund fish samples for the identification of fish species using molecular diagnostics. Ten collected samples were then sent to the laboratory services of BFAR-XI laboratory for the accurate identification of skipjack tuna specimens.
Fish specimens were opened and dissected longitudinally from the anus to the lower jaw, using a pair of dissecting scissors to expose the abdominal cavity. Each fish was eviscerated, and the coelomic cavity was carefully inspected for the presence of parasites on the surfaces of the internal organs and musculature.
The fleshy portions of the fish, particularly the hypaxial and epaxial musculature, were thoroughly examined for parasitic infection. These muscle regions represent the principal edible parts of fish and are of particular concern for food safety, as parasites embedded within these tissues may pose potential exposure risks to consumers, especially when the fish is consumed raw or undercooked. The examination process was conducted systematically to ensure comprehensive and accurate detection of acanthocephalan parasites, following established protocols for parasite isolation and identification in fishery products.
Porcine pepsin (0.5% w/v, P7125, Sigma-Aldrich, St. Louis, MO, USA) dissolved in 0.063 M HCl was used for the artificial pepsin digestion. This protocol allows easy separation of L3 larvae from fresh fish muscle tissue, thus facilitating counting of the recovered larvae. This procedure recovered 92% of live parasites from fresh samples, although using frozen samples may result in poor recovery after pepsin digestion.
Acanthocephalan parasites are commonly identified based on their distinct morphological features. Adult acanthocephalans are typically visible to the naked eye as cylindrical, unsegmented worms, often white or translucent, and range from a few millimeters to several centimeters in length, depending on the species. The most characteristic feature was the presence of an eversible spiny proboscis at the anterior end, which was armed with rows of hooks used for attachment to the intestinal wall of the host.
For identification at the genus or species level, acanthocephalans were fixed in 70% ethanol and examined under a optical microscope. Staining with lactophenol blue or acetocarmine can enhance the visibility of internal structures and diagnostic features. The key morphological characteristics evaluated under the microscope included the structure and arrangement of the proboscis hooks, the shape and size of the proboscis receptacle, the morphology and placement of reproductive organs (such as testes and cement glands), and the overall body shape and size. The anterior (proboscis) and posterior (bursa or genital pore) ends were important for taxonomic differentiation.
Following initial visual morphological evaluation, fixed specimens are typically sent to a parasitologist or expert in acanthocephalan taxonomy for verification and further analysis. Although molecular methods are increasingly used to support species identification, morphological assessment remains the primary method for routine identification.
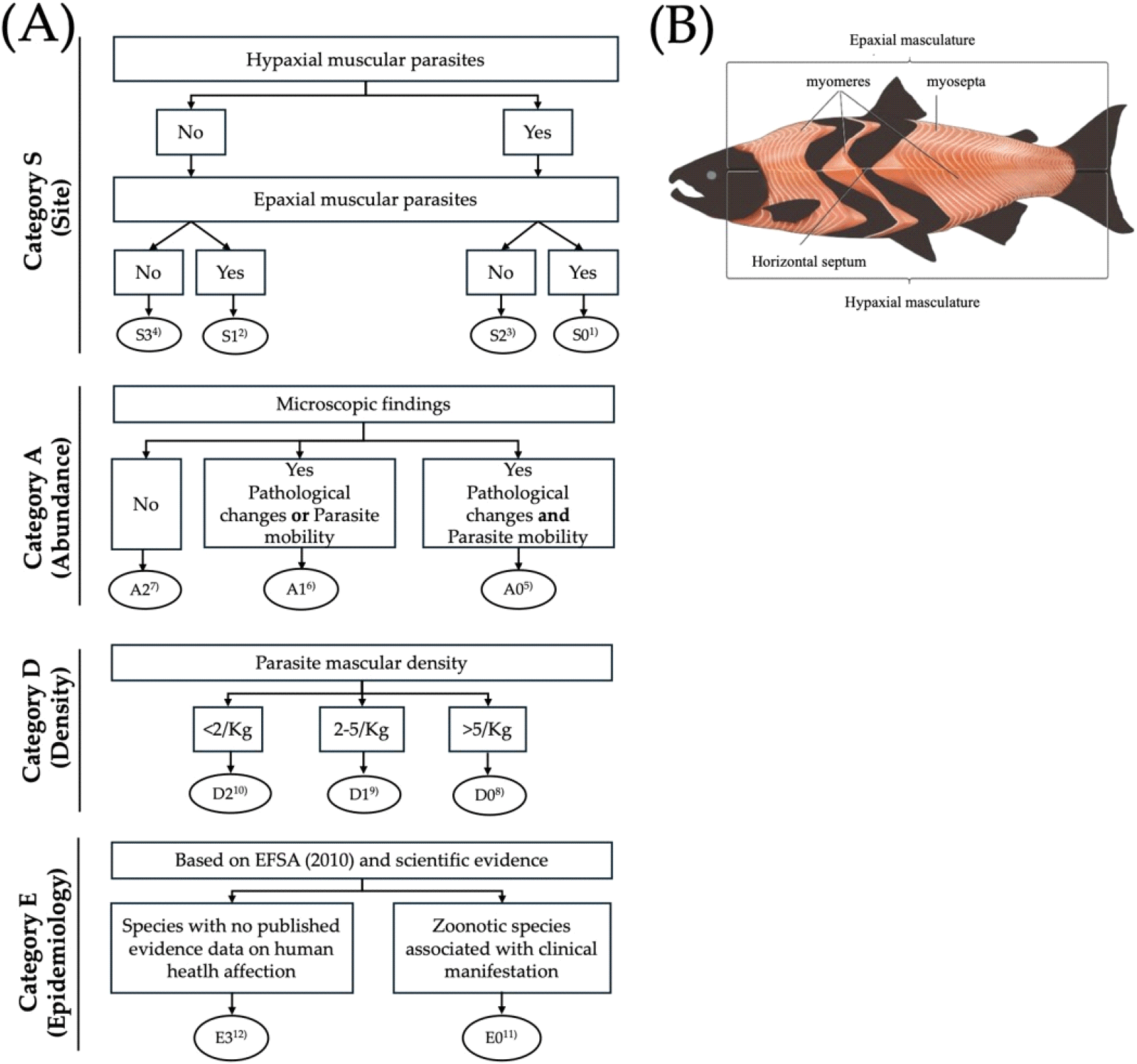
The samples were first grouped according to the following categories: site of parasite (S), Assurance of Quality (A), density (D), and epidemiology (E). These were subsequently divided into alpha-numeric subcategories using a flow diagram (Fig. 2). Category S, where skipjack tuna was scored based on the localization of parasites by observing the number of parasites in the fleshy musculature. Category A included the presence or absence of pathological signs in fleshy portions. Category D includes the number of parasites/kg of fish. Category E for epidemiological relevance of the parasite (E: zoonotic parasite or not).
3. Results and discussion
This study systematically evaluated acanthocephalan parasite infections in 96 skipjack tuna samples from Mati City, Philippines, emphasizing both intestinal and extraintestinal localization, and applied the SADE risk categorization scheme. These findings offer important insights into fisheries management, food safety, and public health, particularly in regions where skipjack tuna is a dietary staple and key economic resource. The 54.17% prevalence of extraintestinal infection underscores the need for enhanced inspection protocols under BFAR Republic Act 10611 freshness standards and may affect compliance with the Codex Alimentarius visual inspection criteria.
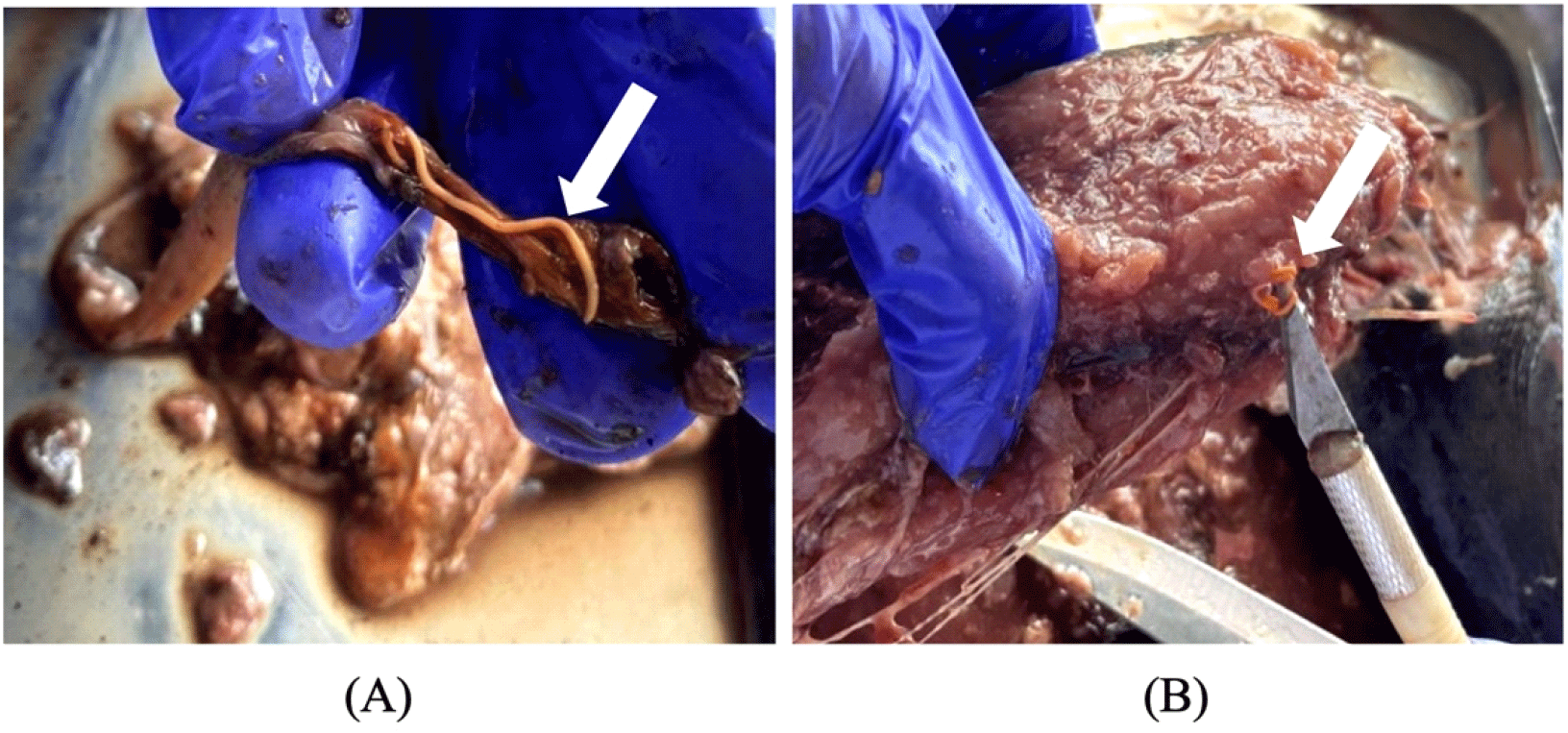
Parasitological assessment revealed significant rates of both intestinal and extraintestinal parasitic infections (Table 1) in the study population. Intestinal parasites were identified in 53 samples, with a prevalence of 55.21%. The total number of intestinal parasites collected was 160, with a mean abundance (MA) of 1.67 parasites per sample (including infected and uninfected samples) and a mean intensity (MI) of 3.02 parasites per infected sample. Extraintestinal parasites were found in 52 samples with a prevalence of 54.17%; across these, 80 parasites were documented, yielding a mean abundance of 0.83 and a mean intensity of 1.54 parasites per infected sample. Gross examination showed visible signs of parasitism (Fig. 3A), while microscopic analysis confirmed the morphological characteristics of the acanthocephalan parasite (Fig. 4).

Intestinal infections by acanthocephalans are well recognized in marine fish and are primarily associated with mechanical damage and inflammation of the gut wall (Alagarsamy Sakthivel, 2020; Bosi et al., 2022; Perrot-Minnot et al., 2023), potentially leading to ulceration or perforation in severe cases (Jithendran et al., 2010). Although not significant zoonotic agents, these parasites compromise fish health and may reduce their market value owing to visible pathology or consumer aversion (Jerônimo et al., 2022; Kvach et al., 2020). They also contaminate processing waters and equipment during gutting/filleting operations, creating cross-contamination risks for subsequent lots under BFAR Good Manufacturing Practices (GMP). The high prevalence observed in this study, consistent with previous reports, reinforces the need for routine inspections aligned with Bureau of Fisheries and Aquatic Resources (BFAR) seafood safety protocols to help prevent potential contamination and ensure product integrity.
A noteworthy finding was the extraintestinal infections, specifically in the hypaxial and epaxial musculature, which are the primary edible portions of the fish. Although less commonly reported, extraintestinal migration raises concerns about high parasite burdens and unique host-parasite interactions that enable tissue penetration. From a market perspective, parasites in muscle tissue may be highly detrimental, especially in raw or lightly processed fish, potentially increasing the risk of rejection during inspection (Kvach et al., 2020). Visible larvae embedded in fillets (Fig. 3B) may contravene BFAR regulations mandating the absence of visible parasites in ready-to-eat portions, potentially triggering port inspection rejections, sashimi-grade failures, retail returns, and economic losses in the Philippine fresh fish markets. These defects pose foreign material hazards during consumption and allergenic cross-contact risks during filleting operations. This underscores the need to integrate BFAR-mandated visual checks with risk-based supply chain traceability and lot-specific parasite logging from capture to processing, and comprehensive surveillance of both intestinal and extraintestinal infection sites. In addition, public education campaigns should be conducted to teach consumers to identify “white spots” in fresh fillets.
SADE scoring of acanthocephalan parasites (Table 2) showed that 47.92% of skipjack tuna samples had no parasites detected in either the hypaxial or epaxial musculature (S3 classification). However, notable parasite presence was recorded in S2 (hypaxial musculature only, 37.50%), S1 (epaxial musculature only, 4.17%), and S0 (both muscle regions, 10.42%). All samples exhibited no visible lesions, discoloration, or tissue damage (A2, 100%). Parasite density was predominantly light (D1, 2-5 parasites/kg, 60.42%) or very light (D2, <2 parasites/kg, 32.29%), with only minor heavy infections (D0, higher than the 5 parasites/kg of fish parasites/kg, 7.29%). According to the epidemiological classification, 78.13% of the detected parasites were E3 (non‑zoonotic species), which implies limited implications for human health and food safety.
The SADE framework provides quantitative risk stratification beyond simple prevalence data. Predominantly low parasite densities (D1/D2, 92.71%), coupled with perfect quality assurance (A2, 100%) and a high proportion of E3 non-zoonotic species (78.13%), collectively demonstrate minimal zoonotic risk and substantiate the overall food safety integrity of the examined samples. This profile generally supports compliance with EU export Regulation (EC) No 853/2004, which requires parasite-free fresh fillets to be obtained via validated freezing or visual certification, alongside BFAR Administrative Order No. 9 s. 2010, mandating lot-specific inspection records, although S0/S2 lots (muscle tissue involvement, 47.92% combined) may increase the risk of non-compliance. Minor heavy-density cases (D0, 7.29%) may increase the risk of rejection under HACCP critical control points and could result in market downgrading from premium fresh/sashimi markets to lower-value cooked products (FAO, 2017).
Operationalizing SADE within BFAR’s National Fishery Products Inspection Program enables risk-based decision trees: S0/S1/S2/D0/D1 lots (parasite-positive) require enhanced candling, transillumination, or molecular screening before release, whereas S3/A2/D2 batches qualify for streamlined processing. This targeted approach optimizes resource allocation and supports the Philippine National Standard PNS/BAFPS 46:2009 for sustainable quality assurance of tuna.
Morphological identification remains reliable for acanthocephalan taxonomy (Schmidt and Roberts, 1989); however, species overlap necessitates molecular confirmation using COI/18S rRNA to differentiate potential zoonotic strains (Perrot-Minnot et al., 2023). Regional consistency with Indo-Pacific baselines validates these Mati City findings (Bosi et al., 2022); however, sampling at a single landing site over a two weeks period during the April dry season warrants expanded surveillance across Davao Gulf, Visayas, and Mindanao fisheries, accounting for potential seasonal, geographical, and fishing ground variations to establish national prevalence maps that can inform adaptive strategies for management.
4. Conclusions
This study documented a high prevalence of acanthocephalan extraintestinal infection (54.17%) in skipjack tuna from Mati City, characterized by predominantly low infection intensity and the absence of visible pathological lesions or tissue damage. Muscle involvement was observed primarily in the S0-S2 classifications, indicating limited but notable parasite presence in muscle tissues relevant to food quality assessment. Despite the low zoonotic risk associated with the predominance of E3 non‑zoonotic species, these findings highlight the need to integrate SADE scoring into BFAR HACCP programs, enhance inspection and processing controls, and expand national surveillance to safeguard Philippine tuna exports and mitigate potential economic impacts due to product downgrading or rejection, thereby maintaining consumer confidence in this vital dietary and economic resource.
