1. Introduction
The discovery of chemical elements and their derivatives has significantly contributed to improving the quality of human life. However, as the prevalence of these chemicals increased in both environmental and personal applications, the subsequent harmful effects also became more apparent (Adebayo, 2024; Check and Marteel-Parrish, 2013; Park et al., 2024). Among these chemicals, chlorinated styrenes (CSs), also known as polychlorinated styrenes (PCSs), including octachlorostyrene (OCS) (CAS 29082-74-4), were nominated in February 1999 as chemicals of concern under the National Industrial Chemicals Notification and Assessment Scheme (NICNAS, 2002). Moreover, 2-chlorostyrene (2-CS), 3-chlorostyrene (3-CS), and 4-chlorostyrene (4-CS) were likewise selected for further investigation due to growing concerns about their environmental persistence and potential for bioaccumulation (NICNAS, 2002). Styrene dibromide, a halogenated styrene derivative, has been documented to migrate from polystyrene insulation materials into food products (Bendall, 2007). This demonstrates the potential for halogenated styrene compounds to serve as food contaminants through packaging-related exposure pathways. In particular, OCS has been categorized in the Priority 1 group under the Persistent, Bioaccumulative, and Toxic (PBT) chemical program, reflecting a high level of concern for both environmental and human health. Therefore, to manage the risks associated with these chemicals, several regulatory bodies have established occupational exposure limits (Adebayo, 2024; Adebayo, 2025; Jeong, 2024; Lee, 2024; Park, 2024). For example, the American Conference of Governmental Industrial Hygienists (ACGIH) has established exposure limits for 2-CS at a shor-term exposure limit (STEL) of 75 μg/g (15-minute exposure) and a threshold limit value (TLV) of 50 μg/g as a time-weighted average (TWA) over an 8-hour workday and 40-hour workweek (NICNAS, 2002). These standards have also been adopted by the National Occupational Health and Safety Commission (NOHSC) in Australia, which has designated 2-CS as a hazardous substance. The NOHSC lists exposure limits identical to those of ACGIH: 50 μg/g (283 mg/m3) as a TWA and 75 μg/g (425 mg/m3) as a STEL (NICNAS, 2002). Given these concerns, the identification and quantification of trace levels of such hazardous chemicals are essential for accurately characterizing chemical residues in the environment (Kuehl et al., 1981).
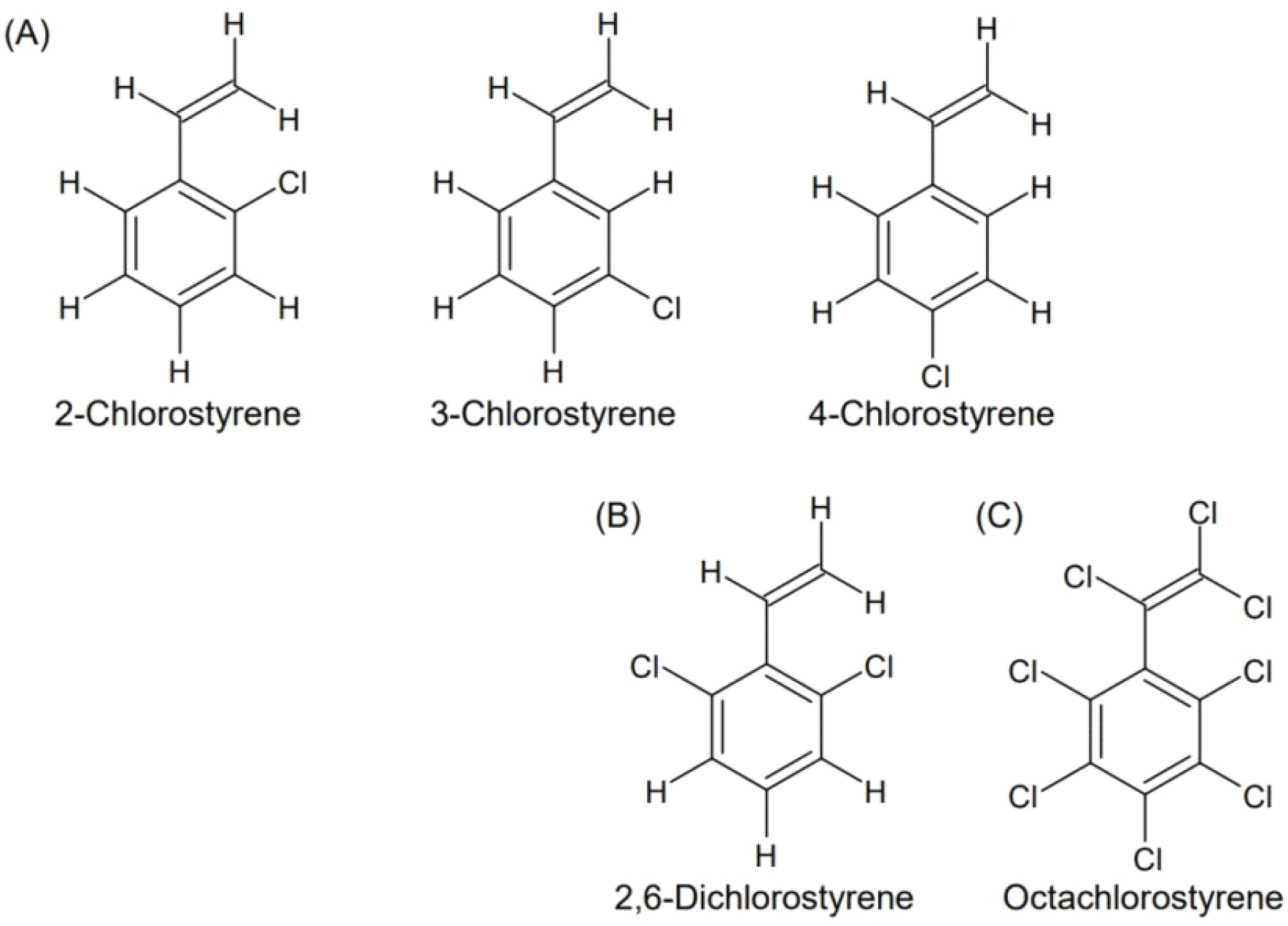
This study aims to provide a comprehensive overview of the formation, toxicity, and analytical methods of chlorostyrenes, with particular emphasis on octachlorostyrene (OCS) as the most environmentally and toxicologically significant congener. The chemical structures of the representative chlorostyrenes discussed in this review are illustrated in Fig. 1.
2. Formation and toxicity
Notably, 2-CS is industrially synthesized as a monomer from 2-ethylchlorobenzene and is often produced alongside 4-CS (NICNAS, 2002). Moreover, 2-CS is primarily used in the production of PCS and for modifying polyesters to enhance flame retardancy and thermal resistance. Additionally, 2-CS is employed in the formulation of various plastics, rubbers, and resins (NICNAS, 2002). Due to the applications od 2-CS, there is a significant risk of environmental release through wastewater from manufacturing or usage sites. Meanwhile, 4-CS is similarly produced via oxidation of p-chloroethylbenzene and commonly co-produced with o-chlorostyrene (NICNAS, 2002). The applications of 4-CS closely mirror those of 2-CS, including its use in PCS synthesis, polyester modification, and CS polymer production.
OCS is not intentionally manufactured for any commercial purpose. Instead, OCS is unintentionally generated during industrial processes involving carbon, chlorine, and high temperatures, often alongside hexachlorobenzene (HCB) and dioxins/furans (NICNAS, 2002). For instance, waste incineration processes can release fly ash containing OCS (Junk and Ford, 1980; Kaminsky and Hites, 1984; King and Sherbin, 1986; NICNAS, 2002). OCS shares structural similarities with HCB (CAS 118-74-1) and is commonly formed under similar conditions. Research has shown that hexachloroethane (HCE)-based aluminum degassing-typically conducted around 740°C, leads to the thermal breakdown of HCE, and the release of releasing chlorine gas. Subsequently, the chlorine gas reacts with molten aluminum to form aluminum chloride, while incomplete degradation of HCE produces persistent by-products such as HCB and OCS. Minor amounts of polychlorinated dibenzo-p-dioxins (PCDDs) and dibenzofurans (PCDFs) are also generated, resulting in a complex emission mixture (Westberg et al., 1997). Other identified sources of OCS include electrolytic chlorine or magnesium production, chlorination and distillation during niobium and tantalum processing, and chlorine fluxing operations in aluminum foundries (NICNAS, 2002). Wastewater from piston manufacturing and aluminum refining represents a significant environmental pathway, particularly during chlorine-based refining and degassing steps (Vogelgesang et al., 1986). Additionally, leachate from landfills containing chlorinated tars and sedimentation basins at aluminum smelters treated with chlorine-based agents have also been implicated as environmental sources of OCS (NICNAS, 2002). According to the NICNAS (2002), OCS formation is closely associated with HCB generation, suggesting the presence OCS in HCB-contaminated waste. Indeed, sediment analyses in affected regions often reveal OCS concentrations above 5 ng/g, which correlate with the presence of three heptachlorostyrene (HpCS) isomers. Furthermore, OCS can be produced during chemical reactions involving chromium (III) oxide (Cr2O3) and carbon tetrachloride (CCl4), as well as in the production of chromium (III) chloride (Mataruse et al., 2010). The unintentional formation of OCS has also been reported in various chlorinated industrial processes such as PVC recycling, aluminum smelting, and metal degreasing using chlorinated solvents (Ren et al., 2022). Additionally, Lahaniatis et al. (1988) reported the formation of OCS during the pyrolysis of various chlorinated hydrocarbons conducted at temperatures of 600-800°C.
CSs, characterized by their high lipophilicity (log Kow 6.86-7.68), are susceptible to biomagnification within the trophic chain. Moreover, CS exhibit binding affinities to both androgen and estrogen receptors, potentially disrupting the homeostasis of thyroid hormones and retinol through their metabolic processes (Carrizo and Grimalt, 2009; Satoh et al., 2001).
Kim et al. (2009) conducted acute toxicity tests for 2CS, 3CS, 4CS, and OCS using algae, daphnia and fish. The experimental results indicated that the EC50 value for 4CS in the freshwater invertebrate *Daphnia magna* was 2.128 mg/L. Similar results were observed in algae and medaka, demonstrating that even compounds with similar structural skeletons can exhibit varying degrees of toxicity depending on the position of their substituents.
Evidence exists of hepatotoxic and nephrotoxic potential associated with MCS exposure (New Jersey Department of Health and Senior Services, 2000).
Chung et al. (2012) demonstrated that 4-CS is more toxic than styrene, and the increased toxicity was closely linked to metabolic activation by CYP2E1, high electrophilicity, glutathione depletion, and covalent binding to intracellular proteins.
Although the International Agency for Research on Cancer (IARC) has not classified 4-CS or 2,6-dichlorostyrene (2,6-DCS) as carcinogenic to humans, these compounds may still induce irritation and exert anesthetic effects on the central nervous system. Meanwhile, current toxicity assessments have been limited to studies involving experimental animals. Direct skin contact with the liquid forms may cause redness and pain, while eye exposure can lead to conjunctival hyperemia, pain, and corneal burns. Comparatively, ingestion may result in symptoms such as abdominal pain and vomiting (Ivan and Ewa, 2000).
Due to the structural similarity of OCS to HCB, OCS is presumed to exhibit comparable toxicological effects (Kim et al., 2009; Seldén et al., 1999). OCS is bioaccumulative and tends to accumulate in animal fat stores, thereby posing potential risks within the food chain (Kypke-Hutter et al., 1986).
Although OCS is not acutely lethal, high doses have been shown to induce symptoms such as reduced mobility and muscle tremors in animal studies. In male rats, a single oral dose of 1,690 mg/kg or higher resulted in increased liver weights, elevated enzymatic activity (e.g., aniline hydroxylase, aminopyrine demethylase), and elevated serum cholesterol and uric acid levels (Chu et al., 1982). In a 28-day dietary study, both male and female rats were fed OCS at concentrations ranging from 0.5 to 500 μg/g. Liver enlargement was observed at ≥50 μg/g, and hepatic microsomal enzyme induction occurred in a dose-dependent manner (≥5 μg/g in males, ≥50 μg/g in females). The highest dose group (500 μg/g) exhibited increased serum cholesterol, total protein, potassium, and sorbitol dehydrogenase levels, accompanied by progressive liver damage (Chu et al., 1982; Chu et al., 1984).
OCS exposure at 5 mg/L caused significant developmental disruptions in Chironomus riparius, with 60% pupation failure, 90% adult emergence failure, skewed sex ratios, larval weight loss, and complete reproductive failure in adults (Lee et al., 2008). Although DNA damage was not observed, OCS upregulated stress response genes, hemoglobin genes, and antioxidant enzymes (Lee and Choi, 2006). A 30-day exposure to 5 mg/L further reduced adult emergence and altered sex ratios.
Chronic OCS exposure induced significant reproductive and behavioral toxicity in Caenorhabditis elegans. In the second generation, egg production dropped to between 3% and 22% of the control levels, and population abundance declined to 6-10%. By the third generation, egg production remained at 30-45%, and locomotion ability was severely impaired (from “+++++” to “++”). On a molecular level, 1,207 genes were differentially expressed in the second generation, with 87 altered in the third generation. Key affected gene families included cytochrome P450 (e.g., Cyp-14A2, was upregulated up to 28-fold), heat shock proteins, and genes involved in hormone signaling and immune response (Kim and Choung, 2009). Meanwhile, Lee and Choi (2009) also observed that chronic OCS exposure, which disrupted adult sex ratios and impaired egg-laying, could severely impact Chironomus populations. While gene expression profiles can act as early warning signals, these profiles alone are not sufficient for comprehensive ecotoxicological assessments.
Ingebrigtsen et al. (1988) found that OCS can induce various hepatic microsomal enzymes such as cytochrome P-450, flavin-containing monooxygenase, glutathione S-transferase, epoxide hydrolase, and UDP-glucuronyl transferase. These findings suggest that OCS may alter the metabolism of both xenobiotics and endogenous compounds such as steroid hormones, potentially leading to reproductive disturbances. Moreover, OCS was shown to enhance the activation of mutagens (e.g., 2-acetylaminofluorene) via liver S9 fractions, indicating a potential cocarcinogenic role. OCS also accumulated in the brains of cod (Gadus morhua) at levels two to four times higher than in rainbow trout (Salmo gairdneri), implying a heightened risk of neurotoxicity in lean fish species. Furthermore, OCS demonstrated very slow elimination rates in both fish and mussels, with residues persisting for 60-90 days post-exposure, underscoring the risk of chronic toxicity due to bioaccumulation.
OCS compounds, such as styrene, undergo biotransformation where epoxide intermediates may form covalent bonds at their site of origin, potentially contributing to liver toxicity. Although direct data on OCS are limited, the structural and toxicological similarity of OCS to HCB suggests a comparable risk profile (Mataruse et al., 2010). OCS was found to induce apoptosis in human liver cell studies via increased reactive oxygen species (ROS) production and caspase-3 activation (Park and Park, 2008). Furthermore, OCS exposure reduced cell viability in a dose- and time-dependent manner, accompanied by significantly elevated ROS levels in HepG2 cells (Ren et al., 2022).
The main findings are summarized in Table 1. In summary, 4-CS exhibits higher acute and cytotoxic effects compared to its isomers, with its toxicity amplified by metabolic activation (CYP2E1). OCS is characterized by its bioaccumulation potential and pronounced chronic, reproductive, and hepatotoxic effects, sharing a mechanism of action similar to hexachlorobenzene (HCB).
3. Analytical methods
To assess the environmental occurrence and potential risks of CSs, monitoring supported by reliable analytical methods is essential. However, such monitoring requires appropriate extraction and purification procedures capable of handling complex matrices. CSs are commonly separated using conventional techniques, such as Soxhlet or ultrasonic extraction; however substantial co-extraction of lipids due to their solubility in organic solvents often complicates the analysis. To minimize matrix interferences, multistep cleanup approaches, including liquid-liquid partitioning, gel permeation chromatography (GPC), and column chromatography, are normally employed. The following sections review validated gas chromatography (GC) techniques, particularly those equipped with mass spectrometry (MS), for the qualitative and quantitative determination of CSs. The analytical methods for the determination of CSs in environmental and biota samples are provided in Tables 2 and 3, respectively.
| Sample | Sample Preparation | Instrunmental analysis | Reference |
|---|---|---|---|
| Water wet sediment | extraction with cyclohexane /isopropanol (1:1), treatment of sulfuric acid, alumina column purification with cyclohexane | GC/ECD | Ofstad et al. (1978) |
| Wet field sediments | Sonication with hexane/acetone (1:1), benzene, Florisil column purification with petroleum ether |
GC/ECD GC/MS |
Pugsley et al. (1985) |
| Wet top sediments | Sonication with methanol, methanol-DCM (1:2), n-hexane extract was rotary vacuum evaporation of n-hexane layer after centrifugation, purification with actives copper. |
GC–EI-MS GC–NICI-MS |
Carrizo et al. (2009) |
| Water | extraction with hexane, cleaned on alumina, Florisil column chromatography with hexane elution |
GC/ECD GC/MS |
Ernst et al. (1984) |
| Wet sediments | Dried for powder, extraction with n-hexane/acetone (2:1) in glass column, treatment of copper |
GC/ECD GC/MS |
Ernst et al. (1984) |
| Wet sediment | Soxhlet-extraction in glass wool thimble with isopropanol and methylene chloride, desulfurization on copper column and fractionation on a 1% water silica gel column, elution with hexane, 9:1 hexane/dichloromethane, dichloromethane, and methanol | GC/MS | Kaminsky and Hites (1983; 1984) |
| Sample | Sample Preparation | Instrunmental analysis | Reference |
|---|---|---|---|
|
Fish Fish fillets (liver) |
Extraction with cyclohexane /isopropanol (1:1), treatment of sulfuric acid, alumina column purification with cyclohexane | GC/ECD | Ofstad et al. (1978) |
| Clam (L. radiata) | Sonication with hexane/acetone (1:1), benzene, Florisil column purification with petroleum ether |
GC/ECD GC/MS |
Pugsley et al. (1985) |
|
Lake trout (S. namaycush) Whitefish (C. clupeiformes) |
Soxhlet-extraction with hexane/acetone (1:1), GPC of SX-2 Bio-Beads followed by silica gel column purification | GC/MS | Swackhamer and Hites (1988) |
| Fish | 10% water silica gel column, extraction with petroleum/dichloromethane (8:2), separation with liquid chromatography on silica gel |
GC/ECD GC/MS |
Steinwandter and Zimmer (1983) |
| Mussels (F. Unionidae) | Soxhlet-extraction with acetone/hexane (59:41), cleanup on column containing Na2SO4, 40% H2SO4 - silica gel and Florisil | GC | Metcalfe and Charlton (1990) |
| Mature breast milk | Extraction with acetone, benzene, purification on Florisil–silicic acid column with 2% dichloromethane in hexane |
GC/ECD GC/MS |
Mes et al. (1986) |
| Fish | Soxhlet-extraction with hexane/ethyl ether (75:25), separation on Micro CelI-E with acetonitrile /acetone (95:5), cleanup on SX-2 GPC column into glass column and Florisil eluted with hexane | GC/MS | Kuehl et al. (1976; 1981) |
| Human venous cord sera | Centrifugation of Serum mixture and recovery standards (TBB, PCB-209), combined n-hexane extract was yielded |
GC–EI-MS GC–NICI-MS |
Carrizo et al. (2009) |
| Planktonic food chain | Water – plankton - mysid (M. relicta) - alewive and smelt – salmonid (O. velinus, S. gairdneri, S. namaycush, S. trutta); Soxhlet-extraction with acetone/hexane, concentration, purification on column with Na2SO4, sulfuric acid-treated silica gel, Florisil, and pretreatment with sulfuric acid |
GC/ECD GC/MS |
Oliver and Niimi (1988) |
| Benthic food chain | Water – sediment – amphipod (P. affinis)/ oligochaete (T. tubifex, L. hoffmeisteri) – sculpin (C. cognatus) – salmonid (O. velinus, S. gairdneri, S. namaycush, S. trutta); Soxhlet-extraction with acetone/hexane, concentration, purification on a column with Na2SO4, sulfuric acid-treated silica gel and Florisil, and pretreatment with mercury |
GC/ECD GC/MS |
Oliver and Niimi (1988) |
| Sprat (C. sprattus) | Extraction with hexane/isopropanol (1:1 v/v), separated hexane layer was treated with sulfuric acid | GC/ECD | Lunde and Ofstad (1976) |
|
Cormorant (P. carbo) Animal species |
Soxhlet-extraction with petroleum ether (40-60°C), purification through a twin-column with dry, partially deactivated alumina and silica gel | GC/MS | de Brauw and Koeman (1973) |
| Fish livers | Cold-extraction with n-hexane, purification with alumina and Florisil column chromatography with n-hexane and n-hexane/ether (1:1). |
GC/ECD GC/MS |
Ernst et al. (1984) |
Quantification of chlorostyrenes in occupational settings is crucial for assessing worker exposure and ensuring compliance with safety guidelines. Analytical methods for this application primarily focus on volatile chlorostyrene compounds, such as monochlorostyrenes and dichlorostyrene isomers, in air samples.
Makhniashvili and Kozieł (2000) developed a specific GC-FID method for the determination of p-chlorostyrene and 2,6-dichlorostyrene in workplace air. Air samples were collected on activated charcoal and fiberglass filters, desorbed with toluene, and separated using a PE-1 capillary column (60 m×0.32 mm i.d., 1 μm film). Their method demonstrated efficacy within a concentration range of 5-500 mg m-3. It achieved high recoveries (≈92-94%), good precision (RSD=0.027 and 0.03, respectively), and a detection limit near 0.05 μg for both analytes.
NIOSH Method 1003 is widely applied for monitoring various halogenated hydrocarbons in workplace air, including chlorinated and brominated styrene compounds. This method involves collecting airborne vapors on activated charcoal tubes, desorbing with carbon disulfide (CS2), and subsequent GC-FID analysis. It provides standardized parameters for sampling conditions, analytical settings, and quality control, and is broadly utilized for evaluating occupational exposure to halogenated volatile organic compounds.
CSs have also been detected in water bodies, necessitating sensitive extraction and analytical techniques. Ernst et al. (1984) identified and quantified OCS in water by extracting samples with n-hexane, followed by purification through alumina and Florisil columns (Ernst et al., 1974; Ernst et al., 1976). Quantification was performed through a Carlo Erba Fractovap 4160 GC with DB-5 quartz capillary columns (30 m, 0.32 mm i.d., 0.3 μm film thickness). OCS was confirmed by GC/MS (Varian Aerograph 2740 coupled to a MAT CH-7) through isotope clustering analysis on a packed column with 3% OV-101 on Chromosorb G-HP. High-resolution GC/MS allowed clear identification of OCS and HpCSs, although PCB isomers can be misidentified as OCS without MS detection.
The analysis of CSs in sediments and soils provides insight into their depositional history and environmental sources. Kaminsky and Hites. (1983; 1984) collected dated sediment cores from Lake Ontario and Soxhlet-extracted core segments in a glass wool thimble sequentially using isopropanol and methylene chloride (24 h each). Extracts were desulfurized on a copper column, fractionated on a 1% water-deactivated silica gel column, and eluted with hexane, 9:1 hexane/dichloromethane, dichloromethane, and methanol. GC/MS analysis were performed using a Hewlett-Packard 5985B operated in methane NCI mode, following initial evaluations in EI mode, with a 30 m DB-5 fused silica capillary column (Jensen et al., 1982). OCS was quantified via its base ion (m/z 308) against external standards, corrected for ∼80% recovery, with RSDs below ±30%. NCI provided the sensitivity to detect hexa- through OCS compounds, which were below the EI detection limits in the sediment extract.
Ernst et al. (1984) also identified and quantified OCS in wet sediments, which were dried and extracted with n-hexane/acetone (2:1) (Ernst et al., 1974; Ernst et al., 1976). Quantification was performed using a Carlo Erba Fractovap 4160 GC with DB-5 quartz capillary columns (30 m, 0.32 mm i.d., 0.3 μm film thickness). OCS was confirmed by GC/MS (Varian Aerograph 2740 coupled to a MAT CH-7) through isotope clustering analysis on a packed column with 3% OV-101 on Chromosorb G-HP. High-resolution GC/MS allowed clear identification of OCS and HpCSs, although PCB isomers can be misidentified as OCS in a packed GC column without MS detection.
Pugsley et al. (1985) examined OCS and PCBs in sediments, which were blended, ultrasonically extracted with hexane/acetone (1:1, v/v) and benzene, and purified on a Florisil column with petroleum ether. Analys was conducted by GC/ECD (Hewlett-Packard 5790A, DB-1 column, 15 m, 0.25 mm i.d.) and GC/MS (Finnigan 4000, 3% OV-1 glass column, 6', 4 mm i.d.), yielding OCS level in surficial sediment of 5.1 μg/kg, with Recoveries of 75-100% over 3-30 μg/kg, and LOQ of 0.2 μg/kg.
CSs accumulate in biota, highlighting the importance of sensitive analytical techniques for biological samples. De Brauw and Koeman (1973) Soxhlet-extracted whole-body homogenates of cormorants and other aquatic species with petroleum ether, followed by twin-column clean-up with partially deactivated alumina and silica gel (Holden and Marsden, 1969). Analysis was performed by GC-MS (Varian Aerograph 1700 coupled to a MAT CH-4) with a 5 ft.×1/8 in. Pyrex glass column packed with 10% DC 200 on Gas-Chrom Q (80/100 mesh) via a silicon membrane separator.
Kuehl et al. (1976) isolated and identified PCSs from fish in the Great Lakes. Here, fish tissues were blended and Soxhlet-extracted with hexane/ethyl ether (75:25). A pesticide-enriched residue without the bulk of lipid was separated from Micro CelI-E by acetonitrile/acetone (95:5) extraction and further cleaned up through a 25×230 mm SX-2 GPC column. Analysis was performed by GC/MS (Varian Aerograph 1740, Varian MAT CH-5) (Kuehl, D.W. et al., 1974), equipped with a 2 m×2 mm glass column packed with 3% OV-101 on an 80/100 mesh Gas Chrom Q.
Lunde and Ofstad (1976) determined chlorinated non-polar compounds in sprat (C. sprattus). Fish samples were homogenized, extracted twice with hexane/isopropanol (1:1 v/v), and the hexane layer was further separated. Lipid extracts were treated with sulfuric acid to isolate chemically persistent compounds. Quantification of HpCS and OCS was carried out by GC/ECD (Perkin-Elmer 3920) with a 2 m × 1/8 in. glass column packed with 3% SE-30 on 80/100 mesh.
Ofstad et al. (1978) determined chlorinated fat-soluble aromatic hydrocarbons, including HpCS and OCS, in fish for human consumption from an area impacted by industrial effluents. Homogenized tissues were extracted using cyclohexane/isopropanol (1:1, v/v), treated with sulfuric acid, and purified by alumina column chromatography before elution with cyclohexane. The sediment and water samples were processed using the same cleanup procedure. Chlorinated compounds were quantified using a Perkin-Elmer 3920 GC equipped with an 3% SE-30 column (6.0 ft×1/8 in.×2.1 mm, stainless steel) and an electron capture detector (ECD).
Building on previous research, Kuehl et al. (1981) assessed OCS and related polychlorinated hydrocarbons in fish from the Great Lakes. Homogenized tissues were packed into a glass column and purified using Florisil, - before being eluted with hexane. Quantification was performed using a Finnigan 4000 GC/MS with SE-30 quartz capillary column (15 m, 0.25 mm i.d.) at MIDs of m/z 378, 380. OCS showed a LOD of 5 ng/g wet weight, exceeding 95% recovery at 100 ng/g fortification. In the MID mode, 226.0 pg injections produced a mean deviation of 6.5% and a standard deviation of 17.9%.
Steinwandter and Zimmer (1983) determined compounds of PCS in Rhine fish. Homogenized fillets were applied to a 10% water silica gel column, and extraction was conducted with on-line cleanup using petroleum/dichloromethane (80:20, v/v). Concentrated extracts were fractionated by liquid chromatography on an activated silica gel. These elution profiles were used for the identification and mass spectrometric characterization of PCSs using GC/ECD and GC/MS with non-extractable bonded DB-1 and DB-5 silica capillary column (30 m, 0.25 mm i.d.).
Ernst et al. (1984) identified and quantified OCS in fish. Homogenized fish livers: OCS was cold-extracted using the same solvent and purified using alumina and Florisil columns with n-hexane and n-hexane/ether (1:1). (Ernst et al., 1974; Ernst et al., 1976). Quantification was performed through a Carlo Erba Fractovap 4160 GC with DB-5 quartz capillary columns (30 m, 0.32 mm i.d., 0.3 μm film thickness). OCS was confirmed by GC/MS (Varian Aerograph 2740 coupled to a MAT CH-7) through isotope clustering analysis on a packed column with 3% OV-101 on Chromosorb G-HP. High-resolution GC/MS allowed clear identification of OCS and HpCSs, although PCB isomers can be misidentified as OCS in a packed GC column without MS detection.
Pugsley et al. (1985) examined OCS and polychlorinated biphenyls (PCBs) in clams, whereby the clam tissues were homogenized and extracted with acetonitrile. Organic extracts from both matrices were purified on a Florisil column with petroleum ether. Analyses were conducted by GC/ECD (Hewlett-Packard 5790A, DB-1 column, 15 m, 0.25 mm i.d.) and GC/MS (Finnigan 4000, 3% OV-1 glass column, 6', 4 mm i.d.). The mean levels of OCS in the clam tissue and surficial sediment were 43.0 μg/kg and 5.1 μg/kg, respectively. Recoveries were recorded as 75-100% over 3-30 μg/kg, with the limits of quantification (LOQ) noted as 0.2 μg/kg, and a chemical concentration factor of 5.9.
Mes et al. (1986) analyzed mature breast milk from Canadian residents for OCS contamination 3-4 weeks postpartum. Milk fat was extracted after centrifugation with acetone and benzene, and purified using a semi-micro Florisil-silicic acid column (Mes and Davies, 1978) eluted with 2% dichloromethane in hexane. Samples were analyzed by Varian 3700 GC on a DB-5 silica capillary column (30 m, 0.24 mm i.d.) and Ni63 ECD, with confirmation by VG ZAB-2F MS at multiple ion detections (MIDs): m/z 378, 380, 382. Background interferences were 0.1-0.4 ng/g, with recoveries ranging from 70% to 106% at 10-30 ng/g fortification.
Swackhamer and Hites (1988) studied the occurrence and bioaccumulation of organochlorine compounds in fish. Homogenized samples were Soxhlet-extracted using hexane/acetone (1:1, v/v) and purified via gel permeation chromatography (GPC) of SX-2 Bio-Beads, followed by silica gel microcolumn cleanup. OCS was analyzed using a Hewlett-Packard 5840 GC with DB-5 (30 m, 0.2 mm i.d.) column and detected by Hewlett-Packard 5985B MS in electron capture negative ionization (ECNI) mode. The detection limit for all compounds was approximately 1 ng/g based on the fish lipid content.
Oliver and Niimi (1988) described analytical methods for the congeners and other chlorinated hydrocarbons of PCB in the Lake Ontario ecosystem, based on an earlier study by Oliver, and Nicol (1982). In brief, samples were Soxhlet-extracted with acetone/hexane, concentrated, and purified on a column containing Na2SO4, sulfuric acid-treated silica gel and Florisil. Biological extracts were pretreated with sulfuric acid, and sediment extracts with mercury to remove interferences. Quantification was conducted using Varian 4600 GC/ECD with dual 30 m capillary columns, DB-5 and DB-17, and confirmed by GC/MS. For OCS, recoveries were around 60-80% using surrogate standards.
In Metcalfe and Charlton (1990), mussels were analyzed for seven organochloride pesticides, including OCS. Biota samples were ground and Soxhlet-extracted with acetone/hexane (59:41), followed by cleanup on an 8mm i.d. column containing Na2SO4, 40% H2SO4-silica gel, and Florisil. Contaminants were analyzed using the Hewlett-Packard 5890 GC with dual 30m capillary columns; HP-5 and HP-17 and the LODs for OCS was 0.01 ng/g wet weight basis, ranging from 2 to 25 g.
Kim et al. (2008) simultaneously analyzed MCSs, DCS and OCS in high-lipid biota. CSs were extracted from fish tissue using ultrasonication with an acetone/n-hexane mixture (5:2, v/v). Lipids were removed via freezing-lipid filtration, followed by purification with Florisil solid-phase extraction (SPE). Quantification was performed by Agilent GC/MS equipped with a DB-5MS column (30 m, 0.25 mm inner diameter, 0.25 μm film thickness) in the electron ionization (EI) mode, employing two internal standards: 13C6-HCB and phenanthrene-d10. Recoveries at 10-100 ng/g were 79.68-95.48% effective with relative standard deviations (RSDs) of 2.96-11.10%, while the limits of detection (LODs) were 0.05-0.1 ng/g (S/n=3), based on two abundant ions.
Carrizo et al. (2009) developed a method to analyze PCSs in human sera. Serum samples were centrifuged and extracted twice with n-hexane. Identification and quantification were performed by an Agilent 6890A GC coupled to an Agilent 5973N MS operated in both electron impact and negative ion chemical ionization (EI/NICI) modes with DB-5 capillary column (60 m, 0.25 mm i.d.). For OCS, the LOD/LOQ value was 0.7/2 ng/g in serum. The NICI analysis demonstrated sufficient sensitivity for CSs in environmental and biological matrices; meanwhile, the EI offered additional spectral data that were useful for qualitative confirmation in samples with high concentrations of these compounds.
4. Occurrence
CSs are bioaccumulative and toxic aromatic compounds produced unintentionally during various industrial processes, including chloro-alkali production and thermal chlorination. CSs are classified as persistent organic pollutants, with several congeners detected in environmental and biological samples worldwide. Among them, OCS is the most widely reported congener due to its high persistence, strong tendency to bioaccumulate, and endocrine-disrupting properties. Detection of CSs in fish, bird eggs, and human blood, particularly in individuals with occupational exposure or high fish consumption, highlights the ecological and human health relevance of this compound class and the industrial origin and potential risks of CS contamination. An overview of studies reporting the Occurence of CSs is summarized in Table 4.
| Country | Sample | Concentration | Reference |
|---|---|---|---|
| Canada | Sediment and clam (L. radiata) from 102 Sites; 9 in Detroit River, 24 in St. Clair River, 69 in Lake St. Clair |
Detroit River; clam OCS (48.7±12.3 ng/g), sediment OCS (1.0±1.2 ng/g), C.F.1) (146.0±102.1 ng/g) St. Clair River; clam OCS (35.9±76.8 ng/g), sediment OCS (14.8±21.4 ng/g), C.F.1) (52.0±46.5 ng/g) Lake St. Clair; clam OCS (43.3±39.6 ng/g), sediment OCS (2.7±4.8 ng/g), C.F.1) (64.0±81.0 ng/g) |
Pugsley et al. (1985) |
| Spain |
Newborn cord sera and maternal sera at 6–8 weeks of pregnancy from Flix (Catalonia, Spain) Top sediments from the Ebro River |
PeCS; 2.1-540 ng/g in sediments, <0.3 ng/L in mothers, <0.3 ng/L in newborns arterial/cord sera HxCS (11,14,15); 2.7-290 ng/g in sediments, <0.7-6.6 ng/L in mothers, <0.7-3.7 ng/L in newborns arterial sera, <0.7-2.2 ng/L in newborns cord sera HpCS (4,5,6); 27.5-610 ng/g in sediments, <0.7 ng/L in mothers, <0.7 ng/L in newborns arterial/cord sera OCS; 66-15,000 ng/g in sediments, 63 ng/L in mothers, 25/17 ng/L in newborns arterial/cord sera |
Carrizo et al. (2009) |
| North sea |
Water from Elbe estuary, Weser estuary, German Bight Sediments from German Bight fish liver2) from A, B, C, D, E, F |
Water: 1.03-1.94 ng/L at Elbe estuary, 0.12 ng/L at weser estuary, 0.03 ng/L at German Bight Sediment: 0.87-1.32 ng/g at German Bight Fish liver: 2.0-164.4 ng/g at A, 2.4-27.2 ng/g at B, <1 ng/g at C, N.D. at D, E, F |
Ernst et al. (1984) |
| North America | Sediment cores from Lake Ontario by the Canada Centre for Inland Waters |
Eastern: <15 ng/g at 1 site (inlet to the Saint Lawrence River), 15-50 ng/g at 2 sites (from Syracuse, NY, through the Oswego River) Midlike: <15 ng/g at 1 site, >100 ng/g at 1 site (offshore of the Genessee River outlet) Western: <15 ng/g at 2 sites, 15-50 ng/g at 2 sites, 50-100 ng/g at 2 sites (from chlorine plants along the shores of the Niagara River) |
Kaminsky et al. (1984) |
| North America | Sediment cores from Lake Ontario, mainly in the Niagara basin from the Niagara River |
HxCSs: 5.9-12 ng/g at 3 sites HpCSs: 0.1-18 ng/g at 6 sites, Tr3) at 1 site OCS: 0.3-49 ng/g at 9 sites, Tr3) at 1 site |
Kaminsky et al. (1984) |
| North America |
Water from Lake Ontario Bottom/suspended sediments from the Lake Ontario/near the Niagara, Mississauga, and Rochester Basins Planktonic/Benthic food chain |
OCS in Water: 4.7±1.3 pg/L OCS in Sediments: 11±5.1 ng/g in bottom, 4.0±1.2 ng/g in suspended OCS in the planktonic food chain4), 0.3 ± 0.05 ng/g in plankton, 0.9±0.7 ng/g in mysids (M. relicta), 14 ng/g in alewife, 5.5±1.9 ng/g small smelts, 9.5 ng/g in large smelts, 44±44 ng/g in salmonids OCS in the benthic food chain5), 6.9±2.9 ng/g in amphipods (P. affinis), 2.5±1.2 in oligochaetes (T. tubifex, L. hoffmeisteri), 16 ng/g in sculpins (C. cognatus), 44±44 ng/g in salmonids |
Oliver and Niimi (1988) |
| Norway | Fish from the Frierfjord; cod (G. morrhua), coalfish (G. virers), whiting (G. merlangus), pollack (G. pollachius), plaice (P. platessa), eel (A. vulgaris), sprat (C. sprattus) |
cod: 6.1-92 μg/g of HpCS, 10-361 μg/g of OCS cod liver: 44-121 μg/g (1975), 2.3-8.2 μg/g (Sept. 1976), 1.4-16 μg/g (Oct. 1976) of HpCS 223-675 μg/g (1975), 12-42 μg/g (Sept. 1976), 16-585 μg/g (Oct. 1976) of OCS cod filet: 26-46 μg/g (May-Sept. 1976), 0.3-24.1 μg/g (Oct.1976) of HpCS 131-169 μg/g (May-Sept. 1976), 3.2-164 μg/g (Oct. 1976) of OCS whiting: 16-23 μg/g of HCS, 47-87 μg/g of OCS Whiting liver: 2.7-24 μg/g of HpCS, 28-256 μg/g of OCS (Oct. 1976) Whiting filet: 0.9-15 μg/g of HpCS, 12-165 μg/g of OCS (Oct. 1976) pollack liver: 1.4-3.7 of HCS, 4-11 of OCS pollack filet: 13-14 of HCS, 7-50 of OCS plaice: 11-18 (Sept. 1975), 1.7-19 (May. 1976) of HpCS 15-18 (Sept. 1975), 21-91 (May. 11976) of OCS eel: 1.9-3.2 (Sept. 1975), 1.4-4.7 (May-Sept. 1976) of HpCS 5.0-6.5 (Sept. 1975), 8-25 (May-Sept. 1976) of OCS Sprat: 3.9-12 of HpCS, 10-40 of OCS |
Ofstad et al. (1978) |
| Norway | Fish from the Eidangerfjord (near the fijord); cod (G. morrhua), pollack (G. pollachius), sprat (C. sprattus) |
cod liver: 2.0-3.8 μg/g of HpCS, 6.2-21 μg/g of OCS, cod filet: 0.7-2.5 μg/g of HpCS, 2.9-3.3 μg/g of OCS pollack liver: 1.4-4.1 of HpCS, 5.1-14 of OCS, pollack filet: 0.8-1.1 of HpCS, 3.1-5.2 of OCS sprat: 0.6-2.3 of HCS, 3.6-6.4 of OCS |
Ofstad et al. (1978) |
| North America | Lake trout (S. namaycush) whitefish (C. culpeaformis) obtained from Siskiwit Lake, Isle Royale, Lake Superior |
OCS in Lake trout: 13 ng/g (SM6) & MED7)), 17 ng/g (LG8)), 32 ng/g (XLG9)), 19 ng/g lipid (mean) OCS in Whitefish: 12 ng/g (SM), 24 ng/g (MED), 13 ng/g (LG), 16 ng/g lipid (mean) |
Swackhamer and Hites (1988) |
| North America | Native mussels (F. Unionidae) from 20 stations in the Ottawa and St. Lawrence Rivers |
upper St. Lawrence River: 0.06 ng/g (L10)) in BP, 0.05 ng/g (E11)) in 29 Lac Saint-Francois: 0.07 ng/g (E) in 42 Lac Saint-Louis: 0.04 ng/g (E), 0.03-0.08 ng/g (L) in 55, 0.17-0.21 ng/g (L) in 59 Lac Saint-Pierre: 0.05 ng/g (E) in 103, 0.05 ng/g (E) in 107, 0.16 ng/g (L) in 112 |
Metcalfe and Charlton (1990) |
| North America | Fish from Great Lakes, also include Lake St. Clair, the Ashtabula River and Fields Brook of Ohio |
Lake Superior Apostle Islands, WI; Copper Harbor, MI, Lake Michigan Grand Traverse Bay, MI, Lake Huron Mackinac, MI: ND12) Lake Huron Saginaw Bay, MI: 2.0-110.7 ng/g Lake Erie Woodtic, MI: 33.8 ng/g Lake Ontario Cape Vincent, NY; Oswego, NY; Stony Island, NY: 28.1-86 ng/g Lake St.Clair Anchor Bay, MI: 227 ng/g Ashtabuka River, OH: NQ13)-405 ng/g Fields Brook, OH: 184-256 ng/g |
Kuehl et al. (1981) |
| Norway | Sprat (C. sprattus) from south-eastern Norway; around Frierfjorden contaminated by chlorinated compounds from one main source. Sprat (C. sprattus) from Norwegian west coast as background |
Frierfjorden: 8.8 μg/g (June 74), 11.2 μg/g (Sept 75) of OCS, 2.9 μg/g (June 74), 4.0 μg/g (Sept 75) of HpCS Eidangerfjorden: 3.8 μg/g of OCS, 1.1 μg/g of HpCS (Sept 75) Ormefjorden: 2.0 μg/g of OCS, 0.6 μg/g of HpCS (Sept 75) Sandefjord: <0.1 μg/g of OCS, <0.02 μg/g of HpCS (Sept 75) Torvik, Romsdal: - of OCS, - of HpCS (Sept 75) |
Lunde and Ofstad (1976) |
C.F., chemical concentration factor, i.e., chemical concentration in clam dry weight/sediment dry weight.
Fish liver: A, North sea, German Bight (south of Helgoland); B, North sea, 55° 30’N:04° 50’E/06° 03’E; C, North Atlantic, Fiskenaes Bank (west of Greenland), D, North Atlantic, Tenerife, local fishery; E, Eastern Mediterranean, Methoni, Greece, local fishery; F, Red sea, Hurghada, local fishery.
Planktonic food chain: Water - plankton (from each major basins) - mysid (M. relicta, from Niagar and Rochester Basin of the lake) - alewive and rainbow smelt (from Vineland, Port Credit) - salmonid (O. velinus, S. gairdneri, S. namaycush, S. trutta from Credit River and Vineland).
CSs were first detected in surficial sediments from Lake Ontario in 1982 (Kaminsky et al., 1983), suggesting that CSs originated from waste by-products (known as “taffy”) following electrolytic chlorine production using graphite anodes. Subsequent analysis of dated sediment cores revealed OCS levels up to 280 ng/g dry weight (Kaminsky and Hites, 1984). Notably, OCS levels above 5 ng/g were consistently accompanied by HpCS isomers, while those below 20 ng/g associated with HxCS, and often with chlorinated PAHs, indicating a common industrial source. The observed concentration increased from the 1940s and declined after the 1970s, following the industrial shift to metal anodes. Locally, the highest values occurred near the Niagara River, highlighting historical deposition and the persistence of CSs in aquatic environments.
Pugsley et al. (1985) evaluated contaminant distributions by analyzing sediment and clam (Lampsilis radiata siliquoidea) samples from 102 sites across Lake St. Clair and the St. Clair-Detroit River corridor. OCS exhibited strong bioaccumulation, with clam concentrations approximately 59 times higher than those in sediment samples, while PCBs in clam tissues were roughly twice the OCS levels. Although sediment levels of OCS and PCBs were generally comparable, OCS levels were notably elevated in the St. Clair River. A moderate but statistically significant correlation (r=0.48; p=0.0001; n=72) was observed between the OCS levels in the sediment samples and clam tissues. The primary source of OCS contamination was linked to historical chlorine production activities along the upper St. Clair River.
Oliver and Niimi (1988) investigated PCB congeners and other chlorinated hydrocarbons, including OCS, in environmental matrices in Lake Ontario. OCS was measured at 4.7±1.3 pg/L in water, and 11±5.1, 4.0±1.2 ng/g (dry weight) in the bottom and suspended sediments, respectively (concentration±standard deviation). OCS and the related hydrophobic organics were markedly higher in the bottom sediments than in the water samples, thereby reflecting the low water solubilities and strong affinity of OCS for particular organic matter. Meanwhile, the co-occurrence with PCBs and other persistent organochlorines suggested that OCS has common sources and similar partitioning behavior within this ecosystem.
Carrizo et al. (2009) investigated PCSs in sediments from areas impacted by emissions from a chlorine-alkali plant. OCS was the predominant congener, with concentrations ranging from 66 to 15,000 ng/g wet weight. Several hexachlorostyrene (HxCS) isomers, such as α-HxCS, E-β-HxCS, and Z-α, β-HxCS, were also detected at levels up to 290 ng/g. To improve detection sensitivity and separate PCS isomers, GC coupled with NICI-MS was used alongside conventional EI-MS. Notably, NICI-MS provided higher sensitivity and resolution, enabling clearer separation and detection of congeners in sediment of the CSs’. These results support the superiority of GC-NICI-MS for environmental biomonitoring of PCSs.
The occurrence of Octachlorostyrene (OCS) in the atmosphere, even in remote regions far from industrial sources, is well-documented and confirms its capacity for long-range environmental transport.
Early investigations identified low levels (≈0.35 pg/m3 medians at 5 sites) in the Arctic (Su et al., 2008) and across Canada (Yao et al., 2008). This work was expanded by Choi et al. (2008), who documented the presence of OCS in both the Arctic (≈1.9 pg/m3) and Antarctica (≈0.24 pg/m3), providing clear evidence of its global distribution. More recently, long-term monitoring (2005-2013) at European high-alpine stations further characterized its persistence at 0.75 pg/m3 (medians) in background air (Kirchner et al., 2016).
While these studies focused on establishing the global distribution of OCS, other research has examined its long-term temporal trends and spatial uniformity. Hites et al. (2022) reported that atmospheric concentrations of both Hexachlorobenzene (HCB) and OCS along the Great Lakes region have remained remarkably stable over the past 20-30 years. They measured geometric means of ∼60 pg/m3 for HCB and ∼0.7 pg/m3 for OCS at five disparate monitoring sites. Notably, the uniformity of OCS concentrations across these sites, regardless of adjacent population or industrial load, and the absence of a downward trend underscore the extreme persistence of OCS in the northern troposphere. This suggests that despite regulatory restrictions on its primary by-product sources, OCS continues to cycle in the environment. This finding highlights the need for targeted sampling and source identification of styrene-based chlorinated pollutants in both occupational and ambient air monitoring programs.
De Brauw and Koeman (1973) analyzed extracts from cormorants (Phalacrocorax carbo), seals (Phoca vitulina), and roach (Leuciscus rutilus) to identify unknown chlorinated compounds. High-resolution with GC/MS revealed a major compound with a molecular ion at m/z 376, consistent with a styrene structure bearing eight chlorine atoms, and two additional compounds at m/z 342, which were tentatively assigned as HpCS congeners. Computerized spectrum subtraction (850 LS system) yielded a deconvoluted spectrum closely matching an authentic OCS standard, conclusively identified its presence in cormorants and seals, and was detected at trace levels in fish.
Lunde and Ofstad (1976) analyzed fat-soluble halogenated organic compounds in sprat (Clupea sprattus) from southeastern Norway, impacted by chlorinated discharges from Frierfjorden. GC/MS identified and quantified PCB, HCB, two isomers of HpCSs and OCS. The total and individual chlorinated hydrocarbon concentrations declined as the distance from the polluted source increased. The highest levels occurred in Frierfjorden, with 16,000 for HCB and 8,800 ng/g for OCS, decreasing in more distant fjords such as Eidangerfjorden and Ormefjorden, and becoming nearly undetectable at the control site. Although absolute concentrations varied, the relative proportions of compounds remained consistent, mirroring those in hexane extracts from activated carbon filters and supporting a common contamination source. Moreover, highly chlorinated compounds, such as OCS and HCB, accumulated more than lower chlorinated analogues, suggesting greater resistance to metabolic degradation and excretion.
Ofstad et al. (1978) investigated chlorinated fat-soluble aromatic hydrocarbons in various fish species from an industrially polluted fjord in Norway. Water and sediment samples were also collected near the discharge point. High level of chlorinated compounds were noted in fish, especially near the effluent source, indicating efficient bioaccumulation. The gadoid species showed particularly high levels of HpCSs and OCS. Those were more enriched in liver than fillet, likely due to the occurrence of slower exchanges in liver tissue. Chromatogram comparisons suggested that there is a relative increase in higher chlorinated compounds in fish, while lower ones appear to be metabolized more rapidly.
OCS was consistently detected in the livers of North Sea fish, with concentrations ranging 2.0-164.4 ng/g wet weight and 25.3-3676.5 ng/g lipid (Ernst et al., 1984). The levels in cod have showed no significant temporal change over the previous decade, indicating sustained exposure. Fish from estuarine areas exhibited higher OCS concentrations compared to those from deeper channels or regions farther from the coast, which were less affected by polluted river inflows. In contrast, OCS was undetectable in fish from less industrialized regions, suggesting that OCS behaves as a persistent, locally accumulated pollutant closely linked to industrial activity, rather than a widely distributed marine contaminant.
Steinwandter and Zimmer (1983) identified 14 isomeric PCS compounds in Rhine fish out of 159 possible congeners. The chromatogram of the extract exhibited a PCB-like pattern, with approximately 50 components showing an elution similar to HCB. Through mass spectra and fragmentography, the peaks were confirmed as PCS compounds. The presence of OCS was also confirmed since OCS exhibited the largest peak in the spectrum. Meanwhile, the spectrum also indicated the presence of other PCS compounds: one pentachlorostyrene (PeCS), eight HxCS and four HpCS isomers. In contrast, lower CSs were not observed in the fish extracts.
Swackhamer and Hites (1988) measured various chlorinated organic compounds in different size classes of lake trout (Salvelinus namaycush namaycush) and whitefish (Coregonus clupeaformis neohantoniensus) from Siskiwit Lake, Isle Royale, Lake Superior. A previous study reported high OCS concentrations in the Lake Ontario ecosystem, attributed to a point source in the Niagara River (Jaffe et al., 1985). The presence of OCS in Siskiwit Lake fish, far from this polluted source, indicates long-range atmospheric transport. Once OCS enters the lake via wet and dry removal processes, OCS enters the food chain and accumulates in fish.
Kuehl et al. (1976) confirmed the presence of PCSs in fish from Lake Ontario, Lake Huron, and the Detroit River. The GC retention times and mass spectral data of the OCS standard, synthesized by two independent methods, were compared with fish extract profiles, which showed the presence of OCS in fish from these sites. The relative retention time (r.r.t) of p,p'-dichlorodiphenyldichloroethylene (p,p'-DDE) ranged narrowly 0.850-0.855 across species, including Stizostedion vitreum and Alosa pseudoharengus, consistent with the OCS standard (r.r.t 0.850). The dominant ion-cluster for OCS appeared between m/z 376 and 386, consistent with its isotope pattern. Fragmentation with chlorine losses produced additional peaks at m/z 341-351 for HpCS and m/z 306-316 for HxCS indicating the presence of several isomers of lower CSs in fish.
In successive studies, Kuehl et al. (1981) investigated OCS and the related polychlorinated compounds in fish from the Great Lakes. OCS was detected in 13 of the 17 samples, at below 5 ng/g in the upper lakes, and up to the 405 ng/g in the lower lakes, representing a 35-40-fold increase in Lake Huron species over four years. Concentrations ranged from 2.0 ng/g in 1977 Lake Huron trout to 405 ng/g in 1979 Ashtabula River northern pike and sucker, with elevated levels also observed in Lake Ontario (up to 281 ng/g) and Fields Brook (up to 256 ng/g). However, while OCS is frequently co-detected with HCB, no consistent correlation was observed. MS identified ions for HpCS (m/z 342/344), HxCS (m/z 308/310), and PeCS (m/z 272/274) isomers, with three HpCS isomers exceeding the noted OCS levels. The bioaccumulation potential of OCS was estimated to be high, with a log bioconcentration factor (BCF) of 4.52, corresponding to ∼33,000-fold enrichment relative to the surrounding water.
Oliver and Niimi (1988) reported the concentration of PCB congeners and various chlorinated hydrocarbons, including OCS, across multiple trophic levels in both planktonic and benthic food chains of Lake Ontario. In the planktonic pathway, OCS increased alongside the trophic level, from 0.3±0.05 ng/g in plankton and 0.9±0.7 ng/g in mysids, to 14 ng/g in alewife, 9.5 ng/g in large smelts, and 44±44 ng/g in salmonids. Similarly, in the benthic chain, levels ranged from 4.0±1.2 ng/g in suspended sediments and 6.9±2.9 ng/g in amphipods to 16 ng/g in sculpins, again reaching 44±44 ng/g in salmonids (concentration±standard deviation, ng/g wet weight). These results provided clear evidence of OCS biomagnification through both pelagic and benthic food webs, which were supported by quantitative data across ecological compartments.
Metcalfe and Charlton (1990) executed biomonitoring for organic industrial contaminants with native mussels (Elliptio complanata and Lampsilis radiata radiata) collected from the St. Lawrence River and Ottawa River. The concentrations of most contaminants identified in mussels from the Ottawa River were 50-75% lower than the lowest values found in the St. Lawrence River. OCS was present at low concentrations in 11 mussels from the St. Lawrence River only, ranging from 0.03 to 0.21 ng/g.
Mes et al. (1986) reported the analysis of 30 chlorinated hydrocarbon contaminants in the breast milk of Canadian women as part of the ongoing monitoring program for HPBs’. A total of 210 samples from five different regions across Canada were analyzed. Although most contaminants were detected in all samples, OCS was less frequently observed. Moreover, while the GC elution patterns exhibited peaks with retention times identical to those of OCS and other compounds, the presence of OCS could not be confirmed by GC/MS analysis.
The OCS concentrations were substantially lower in human serum samples from the same region (Carrizo et al., 2009): 63 ng/g in maternal serum, 25 ng/g and 17 ng/g in newborn arterial and cord sera, respectively. Cord serum exhibited a more diverse isomer profile, including α-HxCS (2.2 ng/g) and several minor HxCS isomers (<0.7 ng/g), indicating potential prenatal exposure. As with the sediment samples, GC-NICI-MS enhanced sensitivity and isomer separation, proving the usefulness of this technique for trace-level detection in biological matrices. These findings highlight the risk of maternal-fetal transfer and in utero exposure to airborne chlorinated pollutants near industrial sources.
5. Conclusions
CSs, especially OCS, have emerged as persistent and bioaccumulative contaminants that have significant implications for both environmental and human health. The presence of chlorinated organic compounds across sediments, fishery organisms, and human samples indicates not only widespread historical deposition but ongoing exposure, underscoring the need for sustained surveillance and effective source control. Thus, understanding the industrial origins and environmental pathways of CSs is essential for designing mitigation and regulatory strategies.
Analytical advancements have expanded the capability to qualify and quantify CSs within complex matrices; however, achieving reliable method validation and optimal sensitivity remains a challenge, underscoring the need for further refinement of high-resolution techniques that can simultaneously capture multiple congeners.
Further research should strengthen toxicological evaluations to clarify ecological and human health impacts and integrate these finding into comprehensive risk assessments. By combining current knowledge on sources, occurrence, and analytical methods, this review provides a framework for addressing critical knowledge gaps and supporting strategies to minimize CS contamination in ecosystems and food chains, ultimately protecting both environmental and public health.
