Introduction
Mackerel, which is found near the western coast of the Pacific and Atlantic area (1), is consumed worldwide. Mackerel is abundant in polyunsaturated fatty acids, which have been proven to actively ameliorate cancers and enhance memory (2). Furthermore, mackerel contains several vitamins and minerals, along with cholesterol, which play important functions in the human body. For example, insufficient consumption of niacin and protein can cause pellagra (3), and minerals participate in energy metabolism to maintain the normal functions of muscles and nerves of the human body (4). Cholesterol is involved in the formation of membranes, and is an important component for the synthesis of bile acid, vitamin D, and steroid hormones (5).
In daily life, fish can be eaten raw, but for better taste, different cooking methods are commonly used, which can have negative or positive effects on the nutritional composition of fish (6). There have already been many studies on the influences of various cooking methods (frying, boiling, steaming, and baking) on the contents of vitamins, mineral, and fatty acids in different types of fish (7,8). Erkan et al. (8) compared the contents of amino acids, vitamins, and other nutrients between raw horse mackerel and that cooked by different methods (frying, grilling, and steaming).
In this study, we analyzed the fat-soluble nutrients (cholesterol, β-carotene, vitamin A, and vitamin E), water-soluble vitamins (vitamin B1, B2, niacin, and folic acid), and selected minerals, along with the proximate composition (moisture, proteins, lipids, and carbohydrates) of mackerel cooked by two different methods, i.e. baking and frying, and compared the effects of the cooking methods on the nutrient composition of mackerel.
Materials and methods
Mackerel was purchased from the local market (Jeonju, Korea) and was cooked as follows. Samples were divided into two groups for baking and frying. For the baking process, 20 g vinegar was added to mackerel (3,980 g), and the surface of mackerel was baked using a grate for 7 min. In the frying process, mackerel (3,846 g) was fried in the presence of soybean oil (577 g) and frying powder (577 g) at 170-180℃ for 57 min. A food blender was used to homogenize each group of samples, followed by dividing into three batches (n=3) for statistical analysis.
Alkaline saponification was performed using the National Laboratory System (NLS) method to obtain non-saponifiable compounds like fat-soluble vitamins and cholesterol (9). Homogenized fish samples (5 g) were weighed and put in 200 mL extraction tubes followed by adding 10 mL of 6% pyrogallol solution (in ethanol). The samples were then vortexed for 2 min and flushed with nitrogen (1 min). Afterwards, the samples were sonicated for 10 min, followed by adding 60% potassium hydroxide (8 mL), and vortexed for further 2 min, and then flushed with nitrogen (1 min). The extraction tubes were placed in a water bath at 75℃ for 1 h at 100 rpm (shaking water bath, BS-21, Lab Companion, Ramsey, MN, USA). The tubes were then washed two times with 10 mL sodium chloride solution (in triple-distilled water, 2%), and an additional 15 mL solution (n-hexane:ethyl acetate, 85:15, v/v, BHT 0.01%) was added for extraction. After vortexing for 2 min, the supernatants were dehydrated through an anhydrous sodium sulfate column, and transferred to other vials. The 15 mL solution was used to extract lipid twice. The supernatants were all collected in a 50 mL volumetric flask. The extra solution was then added to the volumetric flask up to 50 mL and stored at -20℃ until analysis.
Fat-soluble vitamins (A and E) were determined according to the NLS procedure with slight modifications (9). Retinol and DL-α-tocapherol were used as standards for vitamin A and vitamin E quantification, respectively. The HPLC instrument (Yonglin SP930D, Anayang, Korea) for vitamin A determination was equipped with LiChrospher® Diol 100 Column (Merck KGaA, 5 μm, 250 mm×4 mm, Darmstadt, Germany) and a fluorescence detector (Exλ=326 nm, Emλ=470 nm). The mobile-phase contained iso-propanol and n-hexane (5:95 v/v, LC grade) and the flow rate was set to 1 mL/min. Samples (20 μL each) were injected into the column and the analysis was performed for 20 min.
The HPLC instrument for vitamin E determination was equipped with LiChrospher® Diol 100 Column (Merck KGaA, 5 μm, 250 mm×4 mm) and a fluorescence detector (Exλ=290 nm, Emλ=330 nm). The mobile-phase contained n-hexane and iso-propanol (99.4:0.6, v/v) and the flow rate was constant at 1.0 mL/min. Samples (20 μL each) were injected into the column and the analysis was performed for 60 min.
Contents of the vitamin B1, B2, niacin, and folic acid were measured according to the NLS procedure (9).
Cholesterol extraction was performed following the NLS procedure (9). A gas chromatograph (YL 6000 series, Yonglin, Anayang, Korea) was equipped with a YL 6100 instrument fitted with an FID detector. An HP-5 (30 m×0.32 mm, 0.25 μm) column was used to separate the cholesterol, using nitrogen as the carrier gas at a pressure of 18.4 psi and a flow rate of 0.4 mL/min. The temperatures of both the detector and injector were set at 300℃. The oven temperature was set at 285℃ and the analysis time was 15 min. 5α-Cholestane was used as an internal standard for quantification of cholesterol.
β-Carotene content was measured by HPLC according to the NLS procedure (9), using an external calibration curve.
Proximate composition analysis was done with three replicates for measuring moisture, fat, protein, carbohydrate, and crude ash contents following the NLS procedure (9).
Results and discussion
Table 1 shows the contents of fat-soluble vitamins (A and E), cholesterol, and β-carotene in mackerel cooked by different methods. Vitamins A, E, and cholesterol contents of fried fish were found to be 0.010±0.003 mg/100 g, 2.458±0.042 mg/100 g, and 35.361±2.47 mg/100 g, respectively. Compared to baked fish, the contents of vitamin A and cholesterol in fried fish were reduced by 56.52% and 33.40% (p<0.05), respectively. On the other hand, the vitamin E content in fried fish was significantly increased by 275.27% (p<0.05).
The increased vitamin E content in fried fish may be due to the use of frying oil during cooking, because commercial frying oil usually contains additional vitamin E, which might have migrated into the fish tissues.
| Cooking method | ||
|---|---|---|
| Baking | Frying | |
| Vitamin A | 0.023±0.0011)a2) | 0.010±0.003b |
| Vitamin E | 0.655±0.037b | 2.458±0.042a |
| β-Carotene | ND3) | ND |
| Cholesterol | 53.092±1.220a | 35.361±2.470b |
The vitamin A content in fried fish was lower than that in baked fish. Similar results were found in a previous study (10), in which the effect of different cooking methods on the different nutritional qualities of Kutum roach was studied and the content of vitamin A in fried fish was found to be 33.33% less than that in baked fish. Since vitamin A is not very stable at high temperatures, some part of vitamin A may have degraded during cooking. The results shown in Table 1 suggest that the frying process has a more significant impact on reducing vitamin A content than the baking process. Similarly, frying reduced the content of cholesterol more compared to baking (Table 1), partly due to the exudation of fish oil containing lipid-soluble substances like cholesterol. For example, relatively heat-stable and lipid soluble compounds like cholesterol were released from fish tissues to the cooking oil while some of the cooking oil was absorbed into the fish during frying. Thus, lower content of cholesterol is expected in fried fish than in baked fish.
In general, β-carotene is abundant in many vegetables, orange or red fruit chromoplasts (11-12), and some fishes. However, in our study, β-carotene was found in neither baked fish nor fried fish (Table 1).
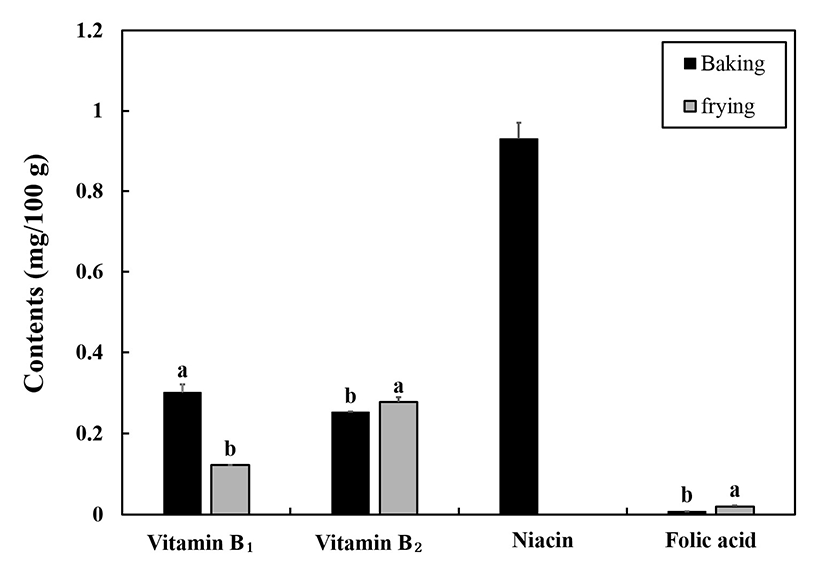
Fig. 1 shows the effect of baking and frying on vitamin B1, B2, niacin, and folic acid contents in mackerel. Compared to baked fish, vitamin B1 content in fried fish decreased by 59.93% (p<0.05). Similar results were observed in study by Erkan et al. (8), in which the lowest vitamin B1 content in horse mackerel was obtained by frying among the different cooking methods (frying, grilling, and steaming). According to the study by Erkan et al. (8), vitamin B1 content decreased from 0.21 mg/100 g in raw fish to 0.042 mg/100 g in fried fish and 0.069 mg/100 g in grilled fish, suggesting that cooking significantly reduced vitamin B1 content and frying led to the maximum reduction in vitamin B1 content. Their results also showed that vitamin B2 contents were reduced from 0.144 mg/100 g in raw fish to 0.136 mg/100 g and 0.109 mg/100 g upon frying and grilling, respectively, suggesting that vitamin B2 may be more stable than vitamin B1 during cooking. In our study, vitamin B2 content in baked fish was significantly lowered compared to that in fried fish (p<0.05) (Fig. 1).
Niacin is also unstable when exposed to high temperatures. Interestingly, niacin was found only in baked fish (Fig. 1) probably due to the different heat exposure conditions (time and temperature) of the different cooking methods. Similar results were obtained by Ersoy and Ozeren (13), who reported that niacin content in raw fish was reduced from 1.13 mg/100 g to 0.04 mg/100 g and 0.73 mg/100 g upon frying and baking, respectively, suggesting that a large amount of niacin is destroyed during the frying process. Meanwhile, the contents of folic acid in both the baked (6.89 μg/100 g) and the fried fish (19.56 μg/100 g) were relatively low compared with the vitamin B1, B2, and niacin contents (Fig. 1).
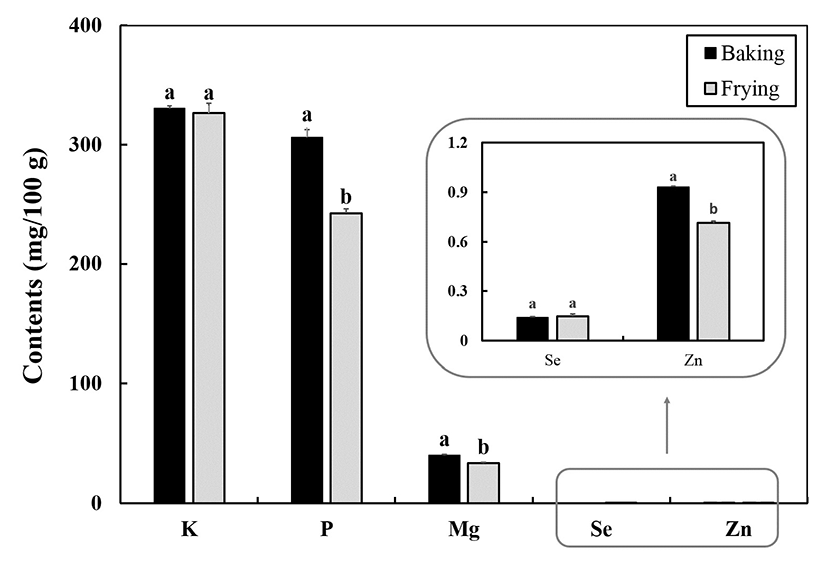
The mineral contents of baked and fried fish samples are presented in Fig. 2. Potassium (K) content was found to be similar in fried fish and baked fish. A similar result was obtained in a previous study on rainbow trout, in which the difference in K content between fried fish and baked fish was not significant (p>0.05) (14). However, P and Zn contents both were found to decrease after baking and frying, with slightly higher levels of P and Zn in baked fish than in fried fish (14). The Mg content of the baked fish was also significantly different from that of the fried one (p<0.05), whereas Se contents of fried and baked fish samples were not significantly different (p>0.05). The effects of baking and frying on the contents of moisture, fats, proteins, carbohydrates, and crude ash are shown in Table 2. While moisture content did not change much, the fat, carbohydrate, and crude ash contents were higher in fried fish than in baked fish.
Furthermore, the content of protein (23.373 g) was found to be higher in baked fish than in fried fish (19.641 g). When frying, part of the water is lost by evaporation, followed by osmosis of frying oil into the fish, resulting in higher fat content in fried fish than in baked fish. Similar results were obtained in a previous study, in which has fat content in fried African catfish increased by 2.17% compared to baked fish (13). Since commercial frying batter usually contains starch and other carbohydrates, fried fish also exhibits higher carbohydrate content than baked fish (Table 2).
